Antibody production
Antibody production
“Antibodies will make a significant contribution to design effective control strategies for COVID-19” – Interview with Prof. Richard Pleass (LSTM)
Intrigued by how parasites are able to elude our immune system, Professor Richard Pleass from the Liverpool School of Tropical Medicine has made remarkable contributions to the design of new immunotherapies, vaccines, and related research tools. By leveraging the Fc region of antibodies, Prof. Pleass and his team continue pushing back the boundaries of knowledge on parasite immunology. ProteoGenix has spoken with Prof. Pleass about his most exciting discoveries and on how they could be applied for the mitigation of the COVID-19 global epidemic.
- ProteoGenix: Can you tell us about your academic and professional journey? What inspired you to devote your career to immunology?
- Throughout your career, you have investigated the use of passive protective immunization against pathogens. What are the advantages and limitations of these approaches in comparison to more conventional treatment and prophylactic methods (e.g. small molecules)?
- Passive immunization against viral pathogens has always been considered more straightforward due to the simplicity of their genomes. However, you’ve also dedicated part of your career in developing passive immunization strategies against parasites. What unique challenges and opportunities have you found in this area and how have you overcome them?
- Would antibody therapy be useful in disrupting the spread of highly contagious and fast progressing epidemics such as Influenza, COVID-19 or Ebola? Why?
- The latest news indicates China is seeking out COVID-19 survivors to use their plasma for convalescent plasma therapy. Nevertheless, there are some conflicting opinions among experts regarding the validity and usefulness of this approach. Do you think plasma therapy could be an effective emergency strategy to counter the progression of this disease? Or should we be focusing our efforts on developing more specific passive immunization strategies?
- Much of your most recent work has been focused on the engineering/mutation of the Fc region of antibodies. Can you summarize how this approach works and what types of methods have been used to modulate glycan content in antibodies?
- In your opinion, why is it worthwhile to pursue in this approach to antibody engineering and what current challenges of modern antibody therapy could be solved by doing so?
- Some experts argue against the development of passive immunization strategies against infectious diseases. In your opinion, what are the greatest challenges hindering research on this topic? What specific challenges associated with antibody production and engineering should we overcome?
- In 2012, you published a highly cited review on the development and future of Fc-fusion proteins [9]. Can you summarize the main findings of this review and tell us how your views have changed since its publication?
- Throughout your career, what has been your most surprising finding?
- Tell us more about other exciting projects happening at your lab.
- What do you think will be the greatest breakthroughs in passive immunization in the coming decades?

ProteoGenix: Can you tell us about your academic and professional journey? What inspired you to devote your career to immunology?
Professor Richard Pleass: I grew up in Zaire (now DRC) in the seventies. My Danish mother was a nurse working at the American embassy and I remember people talking about a monkey disease that a fellow Danish surgeon had died from. The virus in question turned out to be HIV.
As a child, I often overheard dinner table conversations and the surrounding poverty made a huge impression on me. I would accompany my father to the Kinshasa market. He was not interested in shopping for food but in sampling drains for snails that transmit schistosomiasis! I’m sure that is why I chose to study parasites and how our immune systems control them. I can’t help thinking what impact Covid-19 will have on our children’s futures? I hope it inspires some of them to become scientists, nurses and doctors.
Throughout your career, you have investigated the use of passive protective immunization against pathogens. What are the advantages and limitations of these approaches in comparison to more conventional treatment and prophylactic methods (e.g. small molecules)?
I’m a great believer in the power of evolution by natural selection. Antibodies are enormously powerful molecules that have been sculpted by millions of years of evolution to protect us from dangerous organisms that cause disease. The advantage of the antibody molecule lies in its beautiful structure as it can be readily manufactured for high affinity and selectivity, meaning that they are less likely to have unwanted side effects and unexpected safety problems.
In contrast to small molecules, they are also very good at targeting protein-protein interactions that have proved difficult to block by other approaches. They have already been enormously successful in treating cancers and autoimmune disease and more than 550 are now in clinical development. Antibodies, whether they be in diagnostics or therapeutics will make a significant contribution to the design of effective control strategies for COVID-19. At the time of writing they can’t come soon enough!
Passive immunization against viral pathogens has always been considered more straightforward due to the simplicity of their genomes. However, you’ve also dedicated part of your career in developing passive immunization strategies against parasites. What unique challenges and opportunities have you found in this area and how have you overcome them?
Parasites possess unique problems for antibody development. The most important one is that you are faced with an order of magnitude more antigens to choose from than with most viruses. We are only now starting to understand which of these antigens are the best to target with antibodies. Parasites, such as those that cause malaria, also affect neglected populations that are unable to afford the high cost of antibody medicines (some antibodies cost in excess of US$10,000 per month). That said, the technologies for manufacturing antibodies have also developed in pace with their demand. Cost of goods for injected antibodies is now in the range of US$5-25 and numerous published studies have made cogent arguments for their adoption to treat the neglected tropical diseases [1].
Would antibody therapy be useful in disrupting the spread of highly contagious and fast progressing epidemics such as Influenza, COVID-19 or Ebola? Why?
Mass vaccination to raise broadly neutralising antibodies to key antigens is a cost-effective intervention. However, the rapid mutation rate of most viruses means that vaccination e.g. for influenza, has to be repeated annually as antibodies that can neutralise multiple strains and subtypes cannot currently be induced through vaccines. It is too early to say if this is also the case for COVID-19 and Ebola as vaccination campaigns for these two virus types are in their infancy.
The latest news indicates China is seeking out COVID-19 survivors to use their plasma for convalescent plasma therapy. Nevertheless, there are some conflicting opinions among experts regarding the validity and usefulness of this approach. Do you think plasma therapy could be an effective emergency strategy to counter the progression of this disease? Or should we be focusing our efforts on developing more specific passive immunization strategies?
In the absence of effective vaccines and the global over-reliance on a small arsenal of largely ineffective antivirals, past experience with influenza would suggest that convalescent plasma therapy may be useful. Human hyperimmune intravenous immunoglobulin (IVIg) can protect ferrets from the lethal H5N1 and pandemic H1N1 influenza viruses [2], and a small recent double-blind, randomised, placebo-controlled trial in 329 patients with anti-influenza enriched IVIg has shown demonstrable clinical benefit for patients with influenza B over standard care (most commonly Oseltamivir therapy) [3].
Time will tell, but at the time of writing I understand that convalescent plasma therapy has proved useful in Wuhan, and supplies from China are currently being tested in Italy.
Much of your most recent work has been focused on the engineering/mutation of the Fc region of antibodies. Can you summarize how this approach works and what types of methods have been used to modulate glycan content in antibodies?
With a Wellcome Trust Innovator Award [4] we have been making biomimetic replacements of IVIg (see figure below), which is also used for treating autoimmune diseases [5]. These engineered Fcs are based on introducing or removing N-linked glycosylation sites into the IgG1 Fc for changed sialylation profiles [6]. The sialylated fraction of IVIg is believed to confer tolerogenic and inhibitory properties to the injected drug.
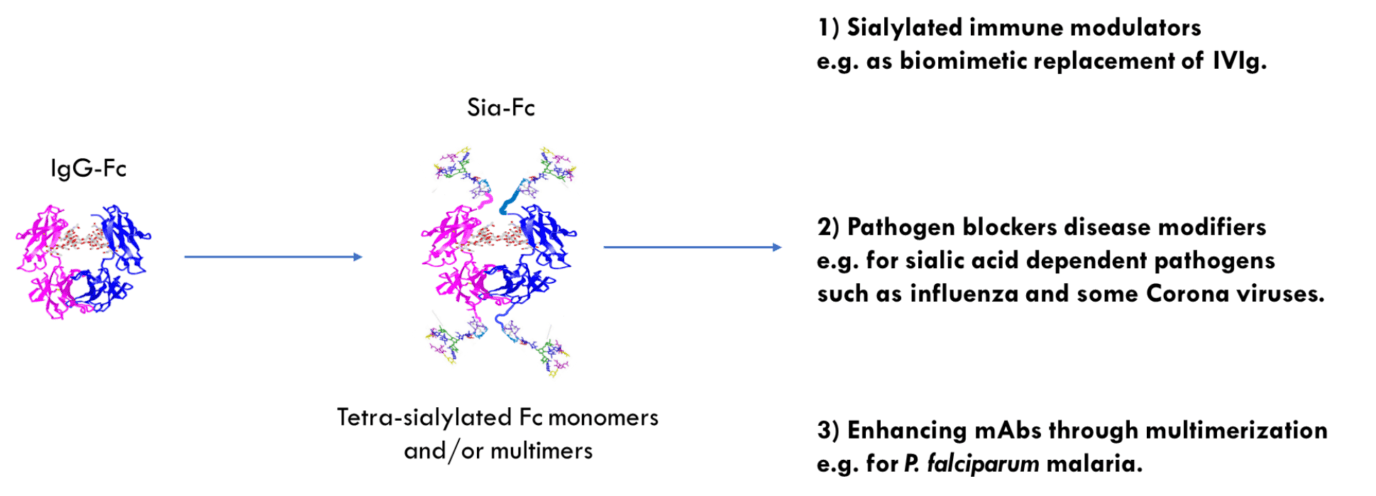
Because neurological disease can also be precipitated by vaccination or infection with influenza, we investigated if our novel molecules could directly interfere with the virus through binding to the prototypic viral sialic acid binding receptor hemagglutinin (HA). We have shown that lead molecules can bind HA from two influenza viruses that diverged approximately 4,000 years ago. As little as 30nM of lead compound inhibited agglutination of human erythrocytes in the WHO standard hemagglutination inhibition assay used to determine the potency of antibodies raised by vaccination. Remarkably, leads molecules were approximately eight-fold more effective than either polyclonal anti-influenza HA or IVIg alone.
The molecules may also protect from other pathogens that are inherently reliant on sialic-acid mediated invasion pathways e.g. some coronaviruses e.g. MERS-CoV [7], adenoviruses, Newcastle Disease virus, and Neisseria meningitidis. They may also block the sialic acid-dependent interaction of group B streptococci with influenza, thereby preventing secondary bacterial infections that occur in seasonal and pandemic outbreaks that contribute significantly to influenza-mediated deaths.
In your opinion, why is it worthwhile to pursue in this approach to antibody engineering and what current challenges of modern antibody therapy could be solved by doing so?
The selling point of lead molecules in development for influenza is that they do not bind Fcg-receptors or complement, and therefore would not interfere with neutralising antibodies found in Flu-IVIG or raised by vaccines. There may therefore be commercial interest in exploring co-formulations of sialylated Fcs with Flu-IVIG. Importantly, certain molecules may also mimic the anti-inflammatory properties of IVIg by binding inhibitory glycan receptors (including Siglecs), thus may also protect patients from influenza-mediated cytokine storms that ultimately kill the infected individual.
IVIg is known to inhibit pro-inflammatory cytokines including IL-6 [11] which is predictive of a poor clinical outcome in COVID-19 patients [12]. In Italy, clinicians have had some success with the anti-IL-6 mAb toclizumab [12] and we are gearing up to investigate if some of our sialylated Fcs can also inhibit IL-6.
Additional advantages over existing approaches are.
- Unique multi-modal mechanism of action (blocks infection and prevents disease)
- Reduced risk of escape mutants (glycan-dependent & smaller molecular footprint than conventional mAbs)
- Increased avidity binding not seen with smaller molecules in development (lead Fcs are tetravalent with respect of glycan binding)
- Fc is therapeutically proven e.g. against linkers or other sialylated scaffolds that may be immunogenic or that have never been tested in humans
- Sialylated Fc has broad therapeutic utility e.g. in treating autoimmune disease
- Sialylated Fc increases in vivo half-life (via enhanced binding to FcRn)
- Fc may be administered through multiple routes (e.g. intra nasal and i.v., s.c.)
- Facile to manufacture in systems that are pharmaceutically proven
- Fc has excellent CoG profiles
Some experts argue against the development of passive immunization strategies against infectious diseases. In your opinion, what are the greatest challenges hindering research on this topic? What specific challenges associated with antibody production and engineering should we overcome?
Given the importance of glycans to the beneficial therapeutic properties of IgG more work is needed to understand the exquisite role played by glycans and their receptors. For example, we have recently found that matched Fc molecules expressed by CHO or HEK cell lines can have profoundly different functional attributes with respect of interaction with either human or pathogen glycan receptors [8].
In 2012, you published a highly cited review on the development and future of Fc-fusion proteins [9]. Can you summarize the main findings of this review and tell us how your views have changed since its publication?
In this review we described interesting avenues for scientific exploration of the IgG1-Fc, including their use for delivering novel drugs, vaccines and diagnostics. Much has happened in the eight years since Dan and I wrote that article, and you have kindly reminded me that I should write an update.
One of the new topics I will be including is recent developments in multimerization technologies to enhance the power of monoclonal antibodies. For example, Genmabs HexaBody® technology to bring enhanced complement-mediated killing to tumour directed mAbs. There are other alternatives to Genmabs multimerization approaches that may yield novel frameworks and concepts for next generation antibodies. These will form the basis for my next review on this important molecule.
Throughout your career, what has been your most surprising finding?
The importance of a good control. That having the correct experimental controls can open up new avenues of research. In 2008, we started exploring the attachment of malaria antigens to multimeric Fcs [10] vaccine delivery, but it was the multimer controls without antigens attached that led our research in its new more exciting direction.
Tell us more about other exciting projects happening at your lab.
The N-linked carbohydrates on our Fcs are amenable to ‘click-chemistry’ and novel material approaches. We are currently exploring how this can be used to further functionalise the Fc for enhanced therapeutics and vaccines. Together with Professor Daniela Ferreira (Head of Clinical Sciences Department at the LSTM) and colleagues in Liverpool we plan to investigate if lead compounds have the potential to control COVID-19 pro-inflammation in the lung.
What do you think will be the greatest breakthroughs in passive immunization in the coming decades?
My experience to date has taught me that the greatest ideas and developments in science can’t always be predicted. I won’t even try to forecast them as in the coming decades the world will have very different problems (e.g. global warming) and diseases (new COVIDs?) to contend with that will inevitably also change the funding landscape. Oh, and yes… I might also look very silly, if I’m still alive that is.
[1] van Huijsduijnen, R. H. et al. Reassessing therapeutic antibodies for neglected and tropical diseases. PLoS Negl Trop Dis. 2020; 14(1): e0007860. doi: 10.1371/journal.pntd.0007860
[2] Rockman, S. et al. Intravenous Immunoglobulin Protects Against Severe Pandemic Influenza Infection. EBioMedicine. 2017; 19: 119-127. doi: 10.1016/j.ebiom.2017.04.010
[3] Davey Jr, R. T. et al. Anti-influenza hyperimmune intravenous immunoglobulin for adults with influenza A or B infection (FLU-IVIG): a double-blind, randomised, placebo-controlled trial. Lancet Resp. Med. 2019; 7:951-963. doi: 10.1016/S2213-2600(19)30253-X
[4] https://wellcome.ac.uk/funding/people-and-projects/grants-awarded/hypersialylated-fragment-crystallisable-regions-treatment
[5] Blundell, P. A. et al. Engineering the fragment crystallizable (Fc) region of human IgG1 multimers and monomers to fine-tune interactions with sialic acid-dependent receptors. J. Biol. Chem. 2017; 292: 12994. doi: 10.1074/jbc.M117.795047
[6] Blundell, P. A. et al. Insertion of N-Terminal Hinge Glycosylation Enhances Interactions of the Fc Region of Human IgG1 Monomers with Glycan-Dependent Receptors and Blocks Hemagglutination by the Influenza Virus. J. Immunol. 2019; 202: 1595. doi: 10.4049/jimmunol.1801337
[7] Park, Y. J. et al. Structures of MERS-CoV spike glycoprotein in complex with sialoside attachment receptors. Nat Struct Mol Biol. 2019; 26(12):1151-1157. doi: 10.1038/s41594-019-0334-7
[8] Blundell, P. A. et al. Choice of Host Cell Line Is Essential for the Functional Glycosylation of the Fc Region of Human IgG1 Inhibitors of Influenza B Viruses. J. Immunol. 2020; 204: 1022. doi: 10.4049/jimmunol.1901145
[9] Czajkowsky, M. D. et al. Fc-fusion proteins: new developments and future perspectives. EMBO Mol Med. 2012; 4(10):1015-1028. doi: 10.1002/emmm.201201379
[10] Mekhaiel, D. N. A. et al. Polymeric human Fc-fusion proteins with modified effector functions. Sci Rep. 2011; 1:124. doi: 10.1038/srep00124
[11] Issekutz, A.C. et al. Intravenous IgG (IVIG) and subcutaneous IgG (SCIG) preparations have comparable inhibitory effect on T cell activation, which is not dependent on IgG sialylation, monocytes, or B cells. Clin. Immunol. 2015; 160(2):123-132. doi: 10.1016/j.clim.2015.05.003
[12] Mehta P. et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020 Mar 16. pii: S0140-6736(20)30628-0. doi: 10.1016/S0140-6736(20)30628-0