Cart (0 Items)
Your cart is currently empty.
View Products Antibody production
Antibody production
Bispecific antibodies (bsAbs) have emerged as a powerful class of engineered therapeutics, particularly in oncology, due to their ability to simultaneously bind two different targets.
Unlike conventional monoclonal antibodies, bsAbs leverage a more complex bispecific antibody structure, enabling novel mechanisms of action such as immune cell redirection and dual pathway inhibition. This structural versatility has driven the rapid expansion of bispecific antibody formats, each designed to optimize therapeutic performance, stability, and manufacturability.
Advances in antibody engineering and recombinant technologies have been essential in overcoming early limitations, paving the way for a growing number of clinical applications.
Bispecific antibodies are artificial proteins engineered to recognize and bind two different antigens or epitopes simultaneously. This dual specificity allows them to perform functions that are not achievable with traditional monoclonal antibodies.
At the molecular level, the bispecific antibody structure differs from standard IgG antibodies by incorporating two distinct antigen-binding sites, often derived from different parental antibodies. This configuration enables the simultaneous engagement of multiple biological targets.
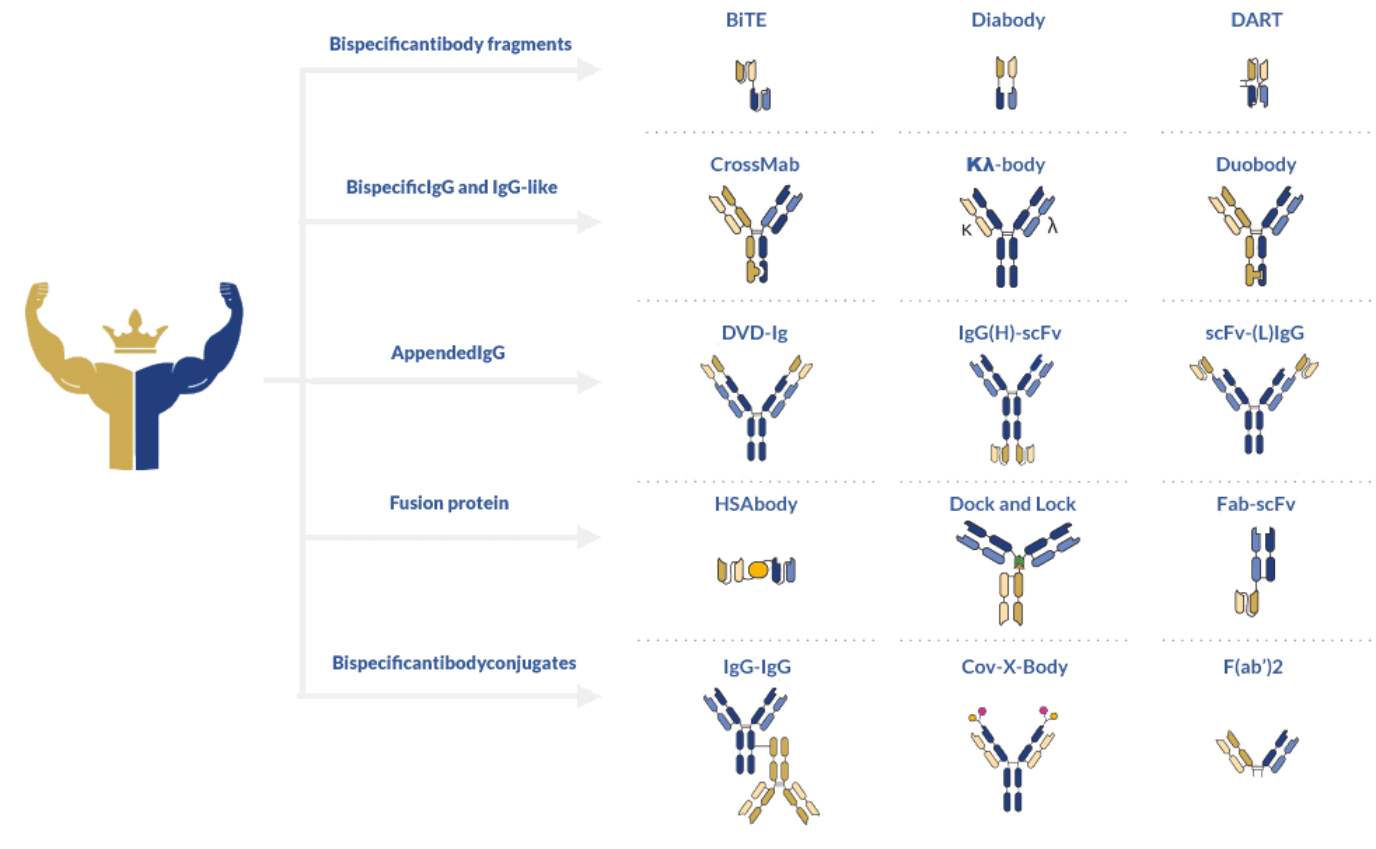
Thanks to the modular nature of antibodies, a wide diversity of bispecific antibody formats has been developed, each offering specific advantages depending on the intended therapeutic application.
Learn more about

Are you working on a bispecific antibody development project?
Discover our bispecific antibody development serviceOne of the most prominent applications of bispecific antibodies is T-cell redirection. These molecules typically bind CD3 on T cells and a tumor-associated antigen, forcing immune cells into close proximity with cancer cells and triggering cytotoxic activity.
Examples of this approach include CD3-targeting bispecific antibodies such as teclistamab and epcoritamab, which have shown strong efficacy in hematological malignancies.
Other mechanisms include:
These mechanisms are directly influenced by the underlying bispecific antibody structure and the spatial arrangement of binding domains, which determine how effectively the molecule can engage its targets.
Explore our therapeutic antibody services
Despite the steady growth of the bispecific antibody therapy market, the immunotherapeutic market is still heavily dominated by conventional monoclonal molecules. However, the double specificity of these man-made molecules makes them more suitable for retargeting (directing an immune cell towards a well-defined target) than conventional antibodies.
Despite the well-known constraints in regard to its production, today, all aspects of a bispecific antibody, independently of their class, can be improved by rational design. Sequence engineering can be used to customize their functionality, enhancing efficiency, and prolonging plasma half-life.
Engineering by rational design is also applied to reduce chain-association issues and improve production yields. For IgG-like bispecific antibodies, common strategies include the “knobs-into-holes” approach that has become the industry’s standard to ensure the correct assembly of two different heavy chains.
In general, bispecific antibodies adhere to a complex architectural classification. Using this system, these antibodies can be divided into fragment-based, asymmetrical, and symmetrical formats.
The first class is defined by the absence of an Fc fragment making it the simplest and easiest to produce of the three classes. Blinatumomab, developed with the BiTE® technology, is the most well-known representative of this class. Other well-known production formats include diabodies and DART (Dual-Affinity Re-Targeting) bispecific antibodies. Diabodies consist of scFv dimers connected by a small peptide linker or covalently linked to each other. In contrast, DART format provides additional stabilization through a C-terminal disulfide bridge.
The lack of the Fc circumvents the chain-association issue and makes bacterial production possible due to the absence of glycans in its structure. Bispecific antibodies of this class can thus be produced very quickly with high yields and reduced costs. However, the lack of Fc also drastically reduces the antibodies’ plasma half-life and abolishes their effector functions.
Of the three main formats, DART has consistently shown to outperform BiTE® in terms of efficiency and plasma half-life. However, more clinical trials are still necessary to determine how these two formats behave under different disease scenarios.
Symmetric formats are quite diverse and differ from the first class by incorporating an Fc fragment. The fragment naturally extends the half-life of these molecules while making their production process more complex. Symmetric bispecific antibodies closely resemble natural antibodies with some differences regarding size and architecture. Most antibodies of this class are tetravalent (2+2). However, the closeness of antigen-binding sites can impair optimal simultaneous binding thus reducing functional valency.
Common bispecific antibody formats of this class include CrossMab, Κλ-body, Duobody, DVD-Ig, IgG(H)-scFv, scFv-(L)IgG, Zybody, among many others.
Asymmetric formats offer a solution to this challenge by increasing the distance between the different antigen-binding sites. However, they also pose additional challenges in terms of chain association that require elaborate engineering.
Bispecific antibodies have gained significant traction in clinical oncology, with multiple approved therapies and a rapidly expanding pipeline.
Their success has been particularly notable in hematological malignancies, while efforts are ongoing to extend their use to solid tumors.
The growing diversity of bispecific antibody formats has played a key role in this progress. Over 100 different formats have been described, ranging from full-length IgG-like molecules to smaller fragment-based constructs, each with distinct pharmacokinetic and functional properties.
These formats allow researchers to tailor:
making bispecific antibodies highly adaptable therapeutic tools.
Despite their potential, bispecific antibodies present several technical challenges:
These constraints require advanced expertise in protein engineering, expression systems, and analytical characterization.
The development of robust workflows integrating antibody design, expression, purification, and validation is essential to ensure successful development.
Bispecific antibody therapies are one of the most versatile tools in our immunotechnology toolbox. For decades, the potential of these molecules has been untapped due to persistent production and stability issues.
This soon may change thanks to the most recent approaches to bispecific antibody engineering approaches that are allowing researchers to orchestrate bispecific antibody assembly at an unprecedented level. In the meanwhile, many of these molecules have proven their therapeutic value in clinical trials. One of the most successful uses of these biotherapeutics rests in the retargeting and activation of important immune cells such as T and NK cells.
Interestingly, many studies have demonstrated that bispecific antibody therapies can find application beyond oncological diseases. Namely in the treatment of infectious diseases or even in regenerative medicine.