Cart (0 Items)
Your cart is currently empty.
View Products


| Size | 100ug, 1MG |
|---|---|
| Isotype | IgG4, kappa |
| Brand | ProteoGenix |
| Product type | Primary Antibodies |
| Clonality | Monoclonal Antibody |
| Expression system | XtenCHO |
| Applications | Elisa, WB |
| Product name | Amlitelimab Biosimilar - Anti-TNFSF4 mAb - Research Grade |
|---|---|
| Source | CAS 2378692-15-8 |
| Species | Humanized |
| Purity | >85% |
| Buffer | PBS buffer PH7.5 |
| Delivery condition | Blue ice (+4°C) |
| Delivery Time | 3-5 days if in stock; 3 week if production needed |
| Storage condition | store at -80°C |
| Brand | ProteoGenix |
| Aliases /Synonyms | Amlitelimab,IMMUNOGLOBULIN G4 (235-PROLINE,242-GLUTAMIC ACID), ANTI-(HUMAN OX40 LIGAND) (HUMAN MONOCLONAL KY1005-2D10 .GAMMA.4-CHAIN), DISULFIDE WITH HUMAN MONOCLONAL KY1005-2D10 .KAPPA.-CHAIN, KY1005, KY-1005, KY1005-2D10, KY-1005-2D10,TNFSF4,anti-TNFSF5 |
| Reference | PX-TA1643 |
| Note | For research use only. Not suitable for clinical or therapeutic use. |
| Isotype | IgG4, Kappa |
| Clonality | Monoclonal Antibody |
Amlitelimab Biosimilar, also known as Anti-TNFSF4 mAb, is a monoclonal antibody that specifically targets the protein TNFSF4 (tumor necrosis factor superfamily member 4). This protein is involved in the regulation of immune responses and its overexpression has been linked to various autoimmune diseases. Amlitelimab Biosimilar is a research grade antibody that is currently being studied for its potential therapeutic applications in the treatment of these diseases. In this article, we will delve into the structure, activity, and potential applications of Amlitelimab Biosimilar.
Amlitelimab Biosimilar is a recombinant humanized monoclonal antibody, meaning it is produced in the laboratory using recombinant DNA technology and has been modified to resemble a human antibody. It is a fully human IgG1 antibody, meaning it belongs to the IgG subclass of antibodies and has a heavy chain of gamma-1 type. The antibody has a molecular weight of approximately 150 kDa and consists of two heavy chains and two light chains. The heavy chains are composed of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains contain one constant domain (CL) and one variable domain (VL). The variable domains are responsible for binding to the target protein, TNFSF4.
Amlitelimab Biosimilar binds specifically to TNFSF4 with high affinity, thereby blocking its interaction with its receptor, OX40. This interaction between TNFSF4 and OX40 is crucial for the activation and proliferation of T cells, which play a key role in autoimmune diseases. By blocking this interaction, Amlitelimab Biosimilar inhibits the activation and proliferation of T cells, leading to a reduction in the inflammatory response. This mechanism of action makes Amlitelimab Biosimilar a potential therapeutic agent for autoimmune diseases.
As mentioned earlier, Amlitelimab Biosimilar is currently being studied for its potential therapeutic applications in autoimmune diseases. Some of the diseases being targeted include rheumatoid arthritis, psoriasis, and multiple sclerosis. In a preclinical study, Amlitelimab Biosimilar has shown promising results in reducing the severity of symptoms in a mouse model of rheumatoid arthritis. It has also been shown to decrease the production of inflammatory cytokines in a human cell model of psoriasis. Additionally, Amlitelimab Biosimilar has been found to have a neuroprotective effect in a mouse model of multiple sclerosis, suggesting its potential use in the treatment of this disease.
In summary, Amlitelimab Biosimilar is a research grade monoclonal antibody that specifically targets TNFSF4, a protein involved in the regulation of immune responses. Its structure, consisting of fully human IgG1 antibody, allows for high specificity and affinity towards its target. By blocking the interaction between TNFSF4 and its receptor, Amlitelimab Biosimilar has the potential to be a therapeutic agent for autoimmune diseases. Further studies are needed to fully understand its efficacy and safety in human clinical trials. However, the current preclinical data shows promising results, making Amlitelimab Biosimilar a potential candidate for the treatment of autoimmune diseases.

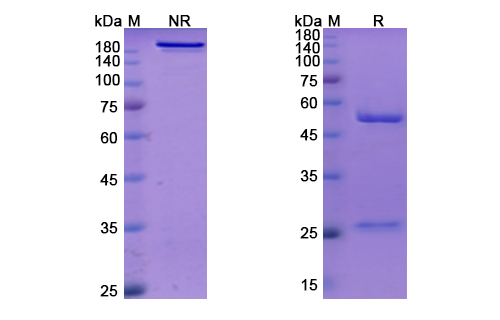
Amlitelimab Biosimilar - Anti-TNFSF4 mAb - Research Grade on SDS-PAGE under reducing and non-reducing conditions. The gel was stained overnight with Coomassie Blue.. The purity of the antibody is greater than 95%.
Related products
Send us a message from the form below
Reviews
There are no reviews yet.