Cart (0 Items)
Your cart is currently empty.
View Products


| Size | 100ug, 1MG |
|---|---|
| Isotype | IgG1, kappa |
| Brand | ProteoGenix |
| Product type | Primary Antibodies |
| Clonality | Monoclonal Antibody |
| Expression system | Mammalian cells |
| Applications | Elisa, WB |
| Product name | Tefibazumab Biosimilar - Anti-Fibrin-binding surface epitope clumping factor A mAb - Research Grade |
|---|---|
| Source | CAS 521079-87-8 |
| Species | Humanized |
| Purity | >85% |
| Buffer | PBS buffer PH7.5 |
| Delivery condition | Blue ice (+4°C) |
| Delivery Time | 3-5 days if in stock; 3-5 weeks if production needed |
| Storage condition | store at -80°C |
| Brand | ProteoGenix |
| Aliases /Synonyms | Tefibazumab,Aurexis,Fibrin-binding surface epitope clumping factor A,anti-Fibrin-binding surface epitope clumping factor A |
| Reference | PX-TA1187 |
| Note | For research use only. Not suitable for clinical or therapeutic use. |
| Isotype | IgG1, kappa |
| Clonality | Monoclonal Antibody |
Tefibazumab Biosimilar, also known as Anti-Fibrin-binding surface epitope clumping factor A mAb, is a research grade monoclonal antibody that has shown promising results in the treatment of various diseases. This article will provide a detailed description of the structure, activity, and potential applications of Tefibazumab Biosimilar.
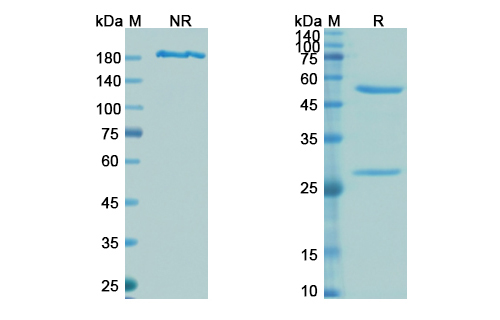
Tefibazumab Biosimilar is a monoclonal antibody that is produced through recombinant DNA technology. It is composed of two heavy chains and two light chains, which are connected by disulfide bonds. The heavy chains have a molecular weight of approximately 50 kDa, while the light chains have a molecular weight of 25 kDa. The antibody has a Y-shaped structure, with two antigen-binding sites located at the tips of the arms.
Tefibazumab Biosimilar targets the surface epitope of clumping factor A (ClfA), a protein found on the surface of Staphylococcus aureus (S. aureus) bacteria. ClfA is a key virulence factor that allows S. aureus to adhere to and invade host cells, leading to infections. Tefibazumab Biosimilar binds to the ClfA protein, preventing its interaction with host cells and inhibiting the growth and spread of S. aureus.
1. Treatment of Staphylococcus aureus Infections S. aureus is a common cause of bacterial infections, ranging from mild skin infections to life-threatening conditions such as sepsis and pneumonia. Tefibazumab Biosimilar has shown promising results in preclinical studies as a potential treatment for S. aureus infections. It has been found to be effective against both methicillin-sensitive and methicillin-resistant strains of S. aureus.
2. Prevention of Biofilm Formation Biofilms are communities of bacteria that are embedded in a protective matrix, making them resistant to antibiotics and the immune system. S. aureus is known to form biofilms, which can cause chronic and recurrent infections. Tefibazumab Biosimilar has been shown to disrupt biofilm formation by binding to the ClfA protein, preventing the bacteria from attaching to surfaces and forming biofilms.
3. Treatment of Skin Infections S. aureus is a common cause of skin infections, such as impetigo, cellulitis, and abscesses. Tefibazumab Biosimilar has been found to be effective in treating these infections, both as a standalone therapy and in combination with antibiotics. Its ability to prevent biofilm formation also makes it a promising treatment for chronic and recurrent skin infections.
4. Potential for Combination Therapy Tefibazumab Biosimilar has shown synergistic effects when used in combination with antibiotics such as vancomycin and daptomycin. This combination therapy has been found to be more effective in treating S. aureus infections than either treatment alone. Tefibazumab Biosimilar’s ability to prevent biofilm formation also makes it a potential adjuvant therapy for antibiotics, as it can increase their efficacy against biofilm-associated infections.
Tefibazumab Biosimilar, also known as Anti-Fibrin-binding surface epitope clumping factor A mAb, is a promising research grade monoclonal antibody that targets the ClfA protein of S. aureus. Its unique mechanism of action makes it a potential treatment for a wide range of S. aureus infections, including those caused by antibiotic-resistant strains. Further clinical trials are needed to fully evaluate the efficacy and safety of Tefibazumab Biosimilar, but it has the potential to become an important therapeutic tool in the fight against S. aureus infections.

Tefibazumab Biosimilar - Anti-clfAClumping factor A mAb - Research Grade on SDS-PAGE under reducing and non-reducing conditions. The gel was stained overnight with Coomassie Blue.. The purity of the antibody is greater than 95%.
Related products
Send us a message from the form below
Reviews
There are no reviews yet.