Introduction
Domvanalimab Biosimilar, also known as Anti-TIGIT mAb, is a promising therapeutic antibody that has shown potential in the treatment of various diseases. This research grade antibody is designed to target TIGIT (T cell immunoreceptor with Ig and ITIM domains), a protein that plays a crucial role in regulating immune responses. In this article, we will discuss the structure, activity, and potential applications of Domvanalimab Biosimilar.
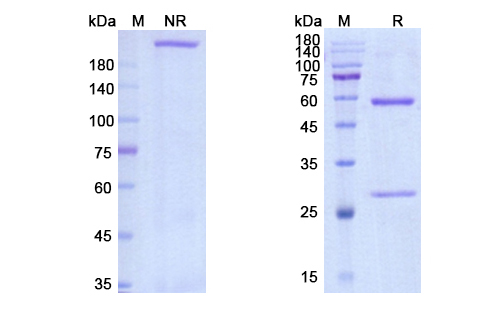
Structure of Domvanalimab Biosimilar
Domvanalimab Biosimilar is a monoclonal antibody, meaning it is produced by a single type of immune cell. It is a fully humanized IgG4 antibody, which means it is derived from human cells and has four subclasses of IgG antibodies. The IgG4 subclass is known for its low binding affinity to Fc receptors, making it less likely to trigger an immune response. This feature is particularly important for therapeutic antibodies as it reduces the risk of adverse reactions in patients.
Activity of Domvanalimab Biosimilar
Domvanalimab Biosimilar works by binding to TIGIT on the surface of T cells. TIGIT is a co-inhibitory receptor that plays a critical role in regulating immune responses. When TIGIT is activated, it inhibits T cell activation and proliferation, leading to the suppression of immune responses. This mechanism is crucial for maintaining immune homeostasis and preventing excessive inflammation. However, in certain diseases, such as cancer, TIGIT can be overexpressed, leading to immune evasion and disease progression. By targeting TIGIT, Domvanalimab Biosimilar can block its inhibitory function and enhance T cell activity, leading to a more robust immune response against cancer cells.
Application of Domvanalimab Biosimilar
Domvanalimab Biosimilar has shown promising results in preclinical studies for the treatment of various cancers, including melanoma, lung cancer, and breast cancer. In these studies, Domvanalimab Biosimilar was able to enhance T cell activity and inhibit tumor growth. Clinical trials are currently underway to evaluate the safety and efficacy of Domvanalimab Biosimilar in cancer patients.
In addition to cancer, Domvanalimab Biosimilar has also shown potential in the treatment of autoimmune diseases. TIGIT has been implicated in the development of autoimmune disorders, and by targeting it, Domvanalimab Biosimilar can potentially modulate the immune response and reduce disease severity. Clinical trials are ongoing to evaluate the efficacy of Domvanalimab Biosimilar in autoimmune diseases such as multiple sclerosis and rheumatoid arthritis.
Conclusion
In conclusion, Domvanalimab Biosimilar is a promising therapeutic antibody that targets TIGIT, a protein involved in regulating immune responses. Its unique structure as a fully humanized IgG4 antibody makes it a safe and effective option for treating various diseases. With ongoing clinical trials, Domvanalimab Biosimilar has the potential to become a valuable addition to the arsenal of therapeutic antibodies for cancer and autoimmune diseases.
There are no reviews yet.