Cart (0 Items)
Your cart is currently empty.
View Products
We brings together technical excellence and tailored support to deliver diagnostic antibodies with guaranteed quality and performance.
Whether you’re developing a diagnostic test in industry or a research assay in academia, you face the same challenges: complex samples, cross-reactivity, lot-to-lot consistency, and final-format performance.
Our custom monoclonal antibody services deliver antibodies with guaranteed quality and performance. They help you achieve reliable results faster and avoid costly re-development during validation.
What derails diagnostic antibody development and how we keep your assay on track
Get a custom diagnostic antibody strategy that tackles the technical and economic challenges below. You reduce re-development risk and keep validation on track.

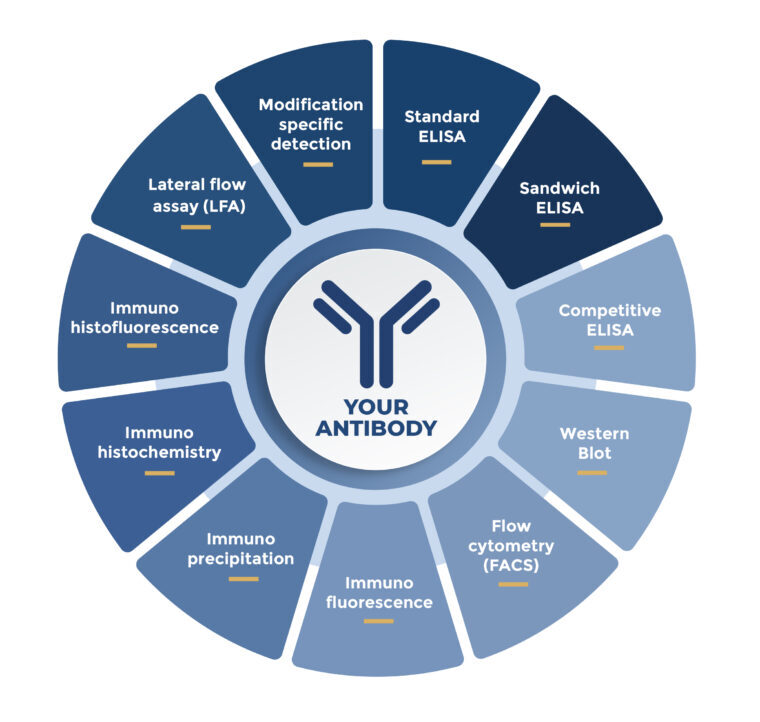
Your antibody is characterized and functionally validated early to de-risk development. Specificity and cross-reactivity are confirmed in relevant complex matrices to keep signals clean in real samples. Targeted characterization speeds candidate comparison before moving to the next stage. Performance is proven in the final assay format to avoid late-stage surprises.

Case report
Case Report: From AI Prediction to Experimental Proof — 85% Signal Reduction with AIxplore® Epitope & Paratope Mapping
With custom recombinant antibody production, programs scale from µg to kg with stable, reproducible quality for confident long-term supply. Batch-to-batch consistency protects data integrity and minimizes re-validation. Format-compatible workflows maintain performance across assays, even for complex targets, while reducing risk to time and budget.
Scale with reproducible quality
Sucess Story
To support EpIeTTX1 pharmacokinetic monitoring, ProteoGenix developed a custom monoclonal antibody capable of specifically detecting the intact therapeutic protein in plasma. With a tailored development strategy and validation under real biological conditions, this project helped TELUM Therapeutics clear a critical development hurdle.
See how we solved a complex pharmacokinetic monitoring challenge
Your end-to-end antibody development project with a single partner
Your project is supported end-to-end to keep every step aligned with the final goal. A dedicated PhD-level expert guides the strategy and execution from kickoff to delivery. Deliverables are IP-free, your antibody can be used without restrictions.
