Introduction
Belantamab Biosimilar is a monoclonal antibody (mAb) that targets TNFRSF17, also known as CD269. It is a research grade antibody with potential therapeutic applications. In this article, we will explore the structure, activity, and potential applications of Belantamab Biosimilar.
Structure of Belantamab Biosimilar
Belantamab Biosimilar is a humanized IgG1 kappa monoclonal antibody. It is composed of two heavy chains and two light chains, each containing variable and constant regions. The variable regions of the antibody are responsible for binding to the target protein, TNFRSF17. The constant regions provide stability and effector functions.
Activity of Belantamab Biosimilar
Belantamab Biosimilar specifically targets TNFRSF17, a transmembrane protein that is expressed on the surface of multiple myeloma cells. TNFRSF17 is a member of the tumor necrosis factor receptor superfamily and plays a crucial role in the survival and proliferation of myeloma cells. By binding to TNFRSF17, Belantamab Biosimilar inhibits its activity and triggers cell death, leading to the destruction of myeloma cells.
Potential Applications of Belantamab Biosimilar
Belantamab Biosimilar has shown promising results in preclinical and clinical studies as a potential treatment for multiple myeloma. It has been found to be effective in both newly diagnosed and relapsed/refractory multiple myeloma patients. In addition, Belantamab Biosimilar has also shown activity against other hematologic malignancies, such as non-Hodgkin’s lymphoma and Waldenström macroglobulinemia.
Antibody Therapy for Multiple Myeloma
Belantamab Biosimilar belongs to a class of drugs known as antibody therapies. These therapies use antibodies to specifically target and attack cancer cells, while sparing healthy cells. This approach offers a more targeted and less toxic treatment option compared to traditional chemotherapy.
Combination Therapy
Belantamab Biosimilar has also shown potential for use in combination therapy with other anti-myeloma agents. Studies have demonstrated that combining Belantamab Biosimilar with other drugs, such as proteasome inhibitors and immunomodulatory agents, can enhance its anti-tumor activity and improve treatment outcomes.
Research Grade Antibody
Belantamab Biosimilar is currently being developed as a research grade antibody, which means it is not yet approved for clinical use. It is being studied in clinical trials to evaluate its safety and efficacy in treating multiple myeloma and other hematologic malignancies. However, the results from these studies have been promising, and it is expected that Belantamab Biosimilar will soon be available for therapeutic use.
Conclusion
In summary, Belantamab Biosimilar is a monoclonal antibody that specifically targets TNFRSF17, a protein expressed on the surface of multiple myeloma cells. By binding to TNFRSF17, Belantamab Biosimilar inhibits its activity and triggers cell death, making it a promising treatment option for multiple myeloma and other hematologic malignancies. As a research grade antibody, it is currently being studied in clinical trials, and it is expected to be available for therapeutic use in the near future.
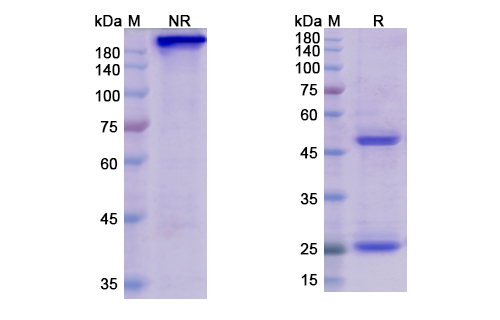
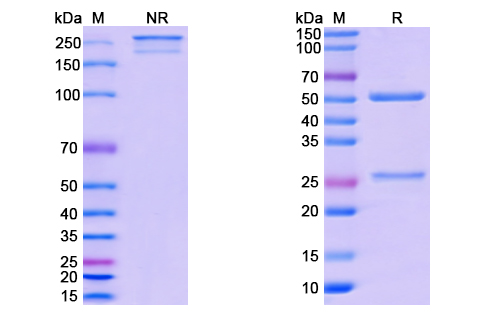
There are no reviews yet.