Cart (0 Items)
Your cart is currently empty.
View Products


| Size | 100µg, 10mg, 1MG, 25mg |
|---|---|
| Isotype | IgG4 |
| Brand | ProteoGenix |
| Product type | Primary Antibodies |
| Clonality | Monoclonal Antibody |
| Expression system | XtenCHO |
| Applications | Elisa, WB |
| Product name | Theralizumab Biosimilar - Anti-CD28 mAb - Research Grade |
|---|---|
| Source | TGN1412, TAB08, CAS: 906068-56-2 |
| Species | Humanized |
| Expression system | XtenCHO |
| Buffer | PBS buffer PH7.5 |
| Delivery condition | Blue ice (+4°C) |
| Delivery lead time in business days | 3-5 days if in stock; 3-5 weeks if production needed |
| Delivery Time | 3-5 days if in stock; 3 week if production needed |
| Storage condition | store at -80°C |
| Brand | ProteoGenix |
| Aliases /Synonyms | anti-CD28, TP44, T-cell-specific surface glycoprotein CD28,TGN1412, TAB08 |
| Reference | PX-TA1927 |
| Note | For research use only. Not suitable for clinical or therapeutic use. |
| Isotype | IgG4 |
Theralizumab Biosimilar, also known as Anti-CD28 mAb, is a monoclonal antibody that targets the CD28 protein on the surface of T cells. This biosimilar is a highly specific and potent therapeutic agent that has shown promising results in various pre-clinical and clinical studies. In this article, we will discuss the structure, activity, and potential applications of Theralizumab Biosimilar.
Theralizumab Biosimilar is a recombinant humanized IgG4 monoclonal antibody, with a molecular weight of approximately 150 kDa. It is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region of the antibody is responsible for binding to the CD28 protein, while the constant region determines the effector functions of the antibody.
The amino acid sequence of Theralizumab Biosimilar is highly similar to the reference product, making it a biosimilar rather than a generic drug. This means that it has comparable structural and functional properties to the reference product, and has undergone rigorous testing to ensure its similarity.
Theralizumab Biosimilar works by binding to the CD28 protein on the surface of T cells. CD28 is a co-stimulatory molecule that is essential for T cell activation and proliferation. By targeting CD28, Theralizumab Biosimilar can modulate T cell responses and regulate immune function.
Studies have shown that Theralizumab Biosimilar can block the interaction between CD28 and its ligands, CD80 and CD86, thereby inhibiting T cell activation. This can be beneficial in conditions where excessive T cell activation is involved, such as autoimmune diseases and organ transplant rejection.
In addition, Theralizumab Biosimilar has also been shown to induce regulatory T cells, which can suppress immune responses and promote immune tolerance. This mechanism of action has potential applications in autoimmune diseases, allergies, and transplant rejection.
Theralizumab Biosimilar has shown promising results in various pre-clinical and clinical studies, and has the potential to be used in a wide range of therapeutic applications. Some of the potential applications of Theralizumab Biosimilar include:
Autoimmune Diseases Theralizumab Biosimilar has shown efficacy in treating autoimmune diseases such as rheumatoid arthritis, psoriasis, and multiple sclerosis. By modulating T cell responses, it can help reduce inflammation and symptoms in these conditions.
Theralizumab Biosimilar has the potential to be used as an immunosuppressive agent in organ transplant patients. By inhibiting T cell activation, it can prevent rejection of the transplanted organ without compromising the overall immune function.
Theralizumab Biosimilar has been shown to be effective in reducing allergic responses, such as asthma and allergic rhinitis. By inducing regulatory T cells, it can suppress the overactive immune response responsible for these conditions.
Theralizumab Biosimilar has also been investigated as a potential treatment for cancer. By targeting the CD28 protein, it can enhance the anti-tumor immune response and potentially improve the efficacy of other cancer therapies.
In summary, Theralizumab Biosimilar is a highly specific and potent monoclonal antibody that targets the CD28 protein on T cells. Its unique mechanism of action has potential applications in various diseases, including autoimmune diseases, organ transplant rejection, allergies, and cancer. With further research and development, Theralizumab Bios

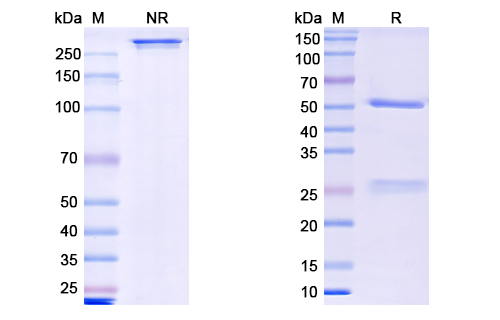
Theralizumab Biosimilar - Anti-CD28 mAb - Research Grade on SDS-PAGE under reducing and non-reducing conditions. The gel was stained overnight with Coomassie Blue.. The purity of the antibody is greater than 95%.
Related products
Send us a message from the form below
Reviews
There are no reviews yet.