About Amivantamab biosimilar
Amivantamab is a bispecific monoclonal antibody with an enhanced FC-part. This antibody is part of the human immunoglobulin G1-kappa type. It is targeting epidermal growth factor receptor (EGFR) and mesenchymal epithelial transition factor (MET, a receptor tyrosine kinase). Both of these factors have key roles in proto-oncogen pathways and invasive growth program.
A treatment by biosimilar recently approved
As Amivantamab biosimilar is targeting proto-oncogen signaling pathway, it is known to have an antineoplasic activity. Janssen Biotech has recently obtained the first approval for RYBREVANT (amivantamab-vmjw –an injection for intravenous infusion) by FDA to treat non-small cell lung cancer (NSCLC) in some conditions. There are also preregistrations in the European Union, Switzerland, Japan, China, Canada and Australia. To be used as a treatment, patients should be adults “with locally advanced or metastatic NSCLC harbouring EGFR Exon 20 insertion mutations whose disease has progressed on or after platinum-based chemotherapy”, according to the FDA.
non-small cell lung cancer: some points
NSCLC is the most common kind of primary type of lung cancer(85% of lung cancers). This cancer is mostly treated using different types of therapy such as immunotherapy and targeted therapy. NSCLCs are carcinomas: this means that the cells developing invasive growth are located on the surface of all the airway parts (bronchi, bronchioles, and alveoli). Symptoms are especially including a chronic cough, chest pain and exhaustion.
A research grade biosimilar
Amivantamab biosimilar is the result of about ten years of research on cross Mab technology. It is produced in mammalian cell line (Chinese Hamster Ovary -CHO) using a recombinant DNA technology. Proteogenix offers this product for research use only.
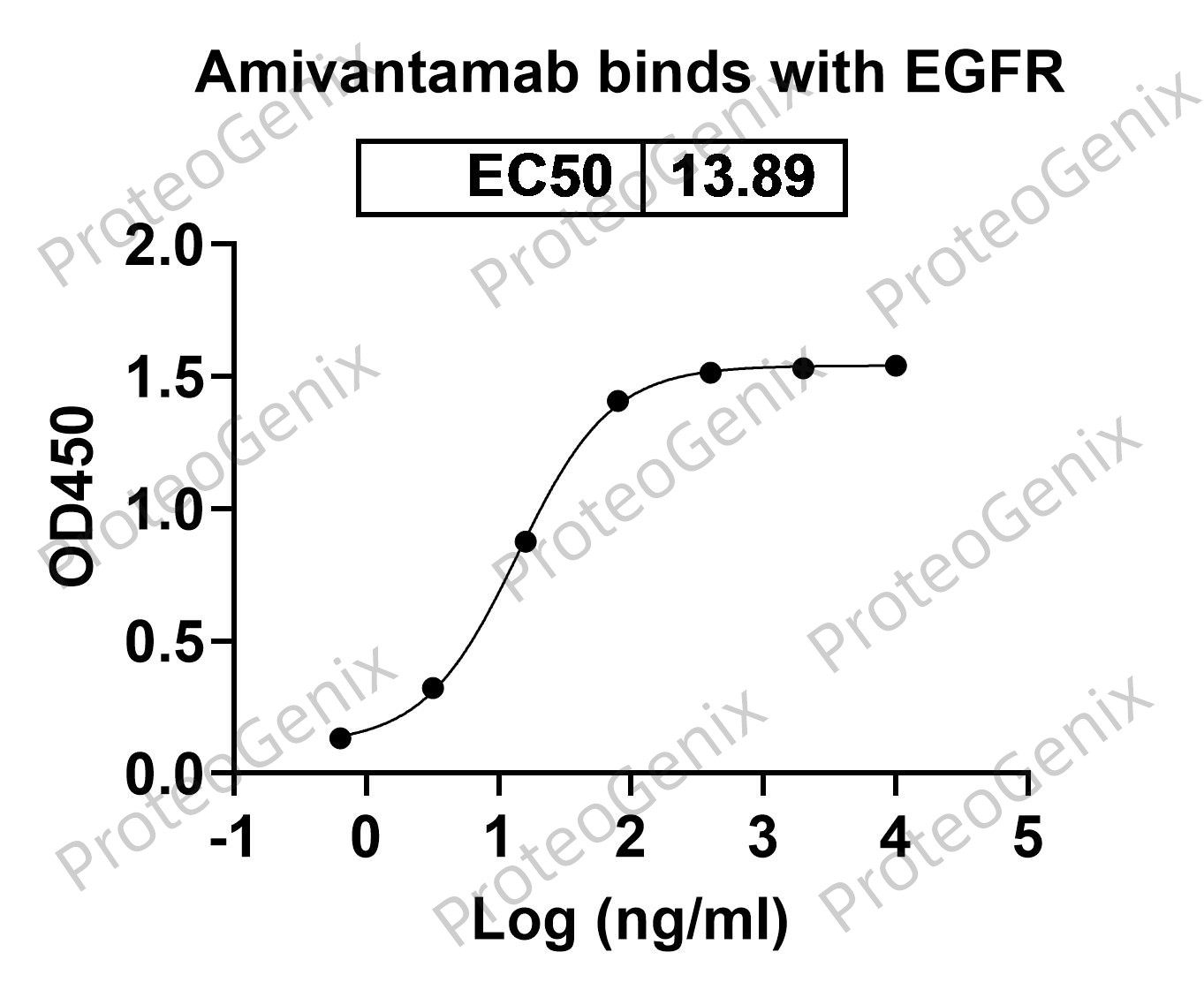
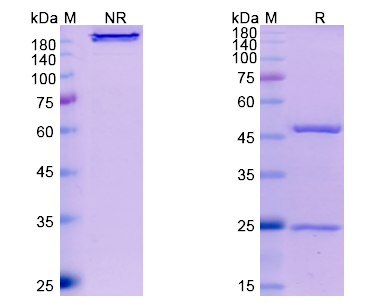
There are no reviews yet.