Briakinumab Biosimilar – Anti-IL12B mAb – Research Grade: A Promising Antibody for Targeting IL12B in Therapeutic Applications Briakinumab Biosimilar – Anti-IL12B mAb – Research Grade: A Promising Antibody for Targeting IL12B in Therapeutic Applications Briakinumab Biosimilar is a monoclonal antibody (mAb) that specifically targets interleukin-12B (IL12B), a cytokine involved in immune response and inflammation. This biosimilar is a highly promising therapeutic agent due to its structural and functional similarities to the approved biologic drug, briakinumab, and its potential for treating various inflammatory and autoimmune diseases.
Structure of Briakinumab Biosimilar
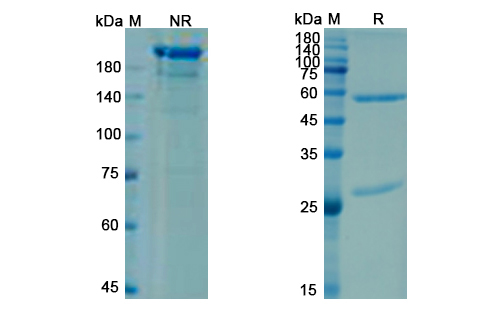
Briakinumab Biosimilar is a recombinant humanized IgG1 monoclonal antibody produced in Chinese hamster ovary (CHO) cells. It consists of two identical heavy chains and two identical light chains, each with a molecular weight of approximately 50 kDa. The antibody has a typical Y-shaped structure with two antigen-binding Fab regions and one Fc region responsible for effector functions.
The amino acid sequence and glycosylation pattern of Briakinumab Biosimilar are highly similar to that of the reference biologic drug, briakinumab. This ensures its comparable safety and efficacy profiles, making it a reliable alternative to the original product.
Mechanism of Action
Briakinumab Biosimilar binds specifically to IL12B, a subunit of the interleukin-12 (IL-12) cytokine, which plays a crucial role in regulating the immune response. IL-12 is primarily produced by antigen-presenting cells and acts on T cells and natural killer cells to induce the production of interferon-gamma (IFN-γ) and other pro-inflammatory cytokines.
By binding to IL12B, Briakinumab Biosimilar prevents the interaction of IL-12 with its receptor, thereby inhibiting the downstream signaling pathways and reducing the production of pro-inflammatory cytokines. This leads to a decrease in inflammation and immune response, making it a potential therapeutic option for various inflammatory and autoimmune conditions.
Applications of Briakinumab Biosimilar
Briakinumab Biosimilar is currently being evaluated in clinical trials for the treatment of psoriasis, psoriatic arthritis, and Crohn’s disease. These conditions are characterized by an overactive immune response, and targeting IL12B with Briakinumab Biosimilar has shown promising results in reducing disease severity and improving symptoms.
Moreover, preclinical studies have also shown the potential of Briakinumab Biosimilar in other inflammatory and autoimmune diseases, such as rheumatoid arthritis, multiple sclerosis, and ulcerative colitis. These findings suggest a broad range of therapeutic applications for this biosimilar in the future.
Conclusion
Briakinumab Biosimilar is a highly promising antibody that specifically targets IL12B, a key cytokine involved in immune response and inflammation. Its structural and functional similarities to the approved biologic drug, briakinumab, make it a reliable alternative with comparable safety and efficacy profiles. With ongoing clinical trials and potential for treating various inflammatory and autoimmune diseases, Briakinumab Biosimilar holds great promise as a therapeutic agent for improving patient outcomes.
There are no reviews yet.