Introduction
Cirevetmab Biosimilar is a novel, research-grade monoclonal antibody (mAb) that specifically targets Transforming Growth Factor Beta 1 (TGFB1) in canines. This therapeutic antibody has shown promising results in pre-clinical studies and has the potential to revolutionize the treatment of various canine diseases.
Structure of Cirevetmab Biosimilar
Cirevetmab Biosimilar is a recombinant, fully humanized IgG1 monoclonal antibody. It is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region of Cirevetmab Biosimilar is engineered to specifically bind to TGFB1, while the constant region is responsible for the effector functions of the antibody.
Mechanism of Action
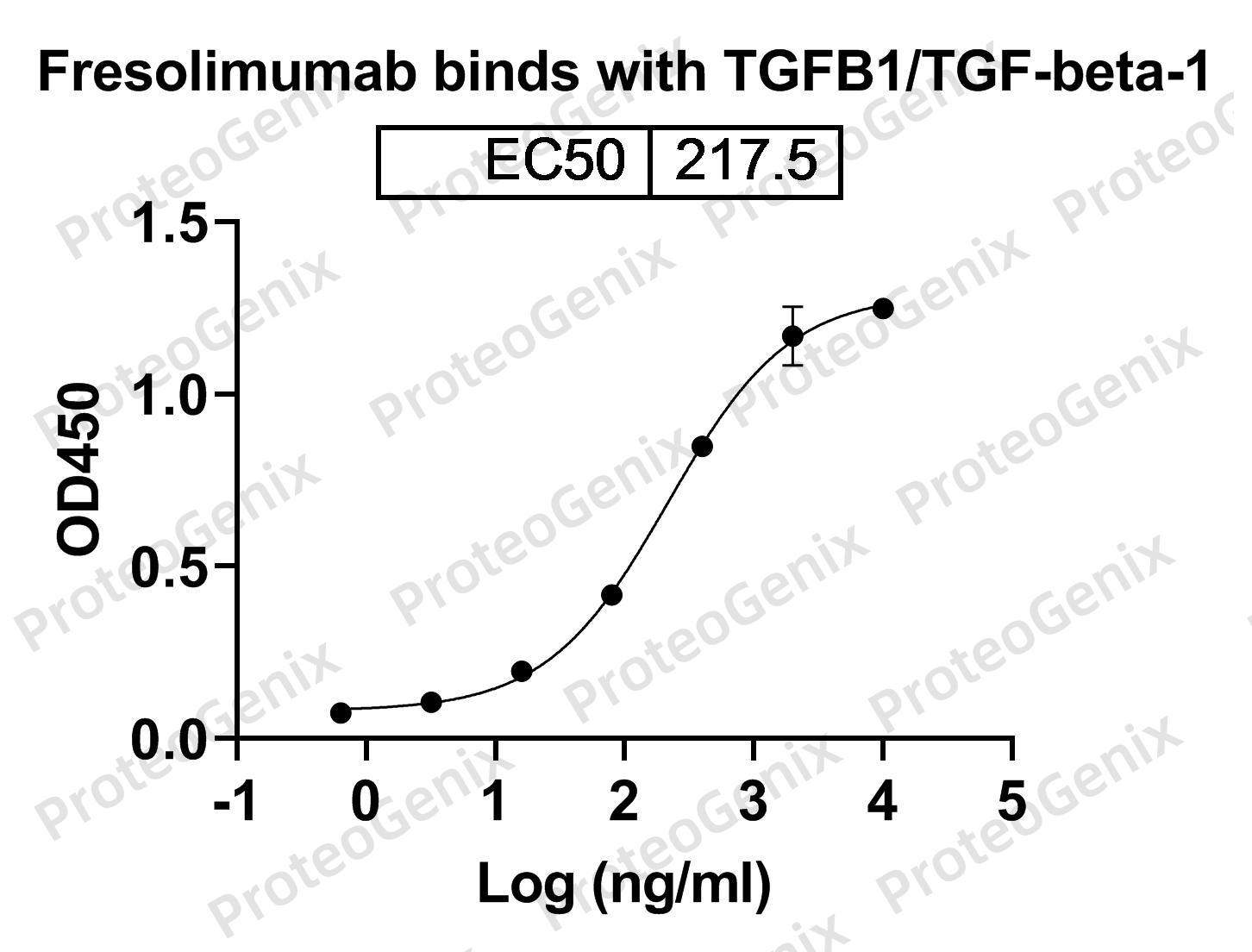
TGFB1 is a cytokine that plays a critical role in regulating various cellular processes, including cell growth, differentiation, and immune response. However, dysregulation of TGFB1 has been linked to the development and progression of several diseases, including cancer, fibrosis, and autoimmune disorders. Cirevetmab Biosimilar binds to TGFB1 and prevents its interaction with its receptors, thereby inhibiting the downstream signaling pathways and reducing the pathological effects of TGFB1.
Applications of Cirevetmab Biosimilar
Cirevetmab Biosimilar has shown promising results in pre-clinical studies for the treatment of various canine diseases, including cancer, inflammatory diseases, and fibrosis. In cancer, Cirevetmab Biosimilar has been shown to inhibit tumor growth and metastasis by blocking the immunosuppressive effects of TGFB1. In inflammatory diseases, Cirevetmab Biosimilar has been found to reduce inflammation and tissue damage by inhibiting the pro-inflammatory effects of TGFB1. In fibrosis, Cirevetmab Biosimilar has been shown to prevent the excessive deposition of extracellular matrix by inhibiting the profibrotic effects of TGFB1.
Advantages of Cirevetmab Biosimilar
Compared to other therapeutic antibodies targeting TGFB1, Cirevetmab Biosimilar has several advantages. Firstly, being a fully humanized antibody, it has a lower risk of immunogenicity and can be administered repeatedly without causing adverse immune reactions. Secondly, Cirevetmab Biosimilar has a high binding affinity and specificity for TGFB1, making it a potent inhibitor of TGFB1 signaling. Thirdly, Cirevetmab Biosimilar has a long half-life, allowing for less frequent dosing and better patient compliance.
Clinical Development of Cirevetmab Biosimilar
Cirevetmab Biosimilar is currently in the pre-clinical stage of development, with promising results in in vitro and in vivo studies. The next step would be to conduct clinical trials to evaluate the safety and efficacy of Cirevetmab Biosimilar in canines. If successful, Cirevetmab Biosimilar could potentially be approved for use in various canine diseases, providing a new and effective treatment option for veterinarians and pet owners.
Conclusion
In conclusion, Cirevetmab Biosimilar is a novel, research-grade monoclonal antibody that specifically targets TGFB1 in canines. Its unique structure and mechanism of action make it a promising therapeutic option for various canine diseases. With further development and clinical trials, Cirevetmab Biosimilar has the potential to improve the lives of countless canine patients and their owners.
There are no reviews yet.