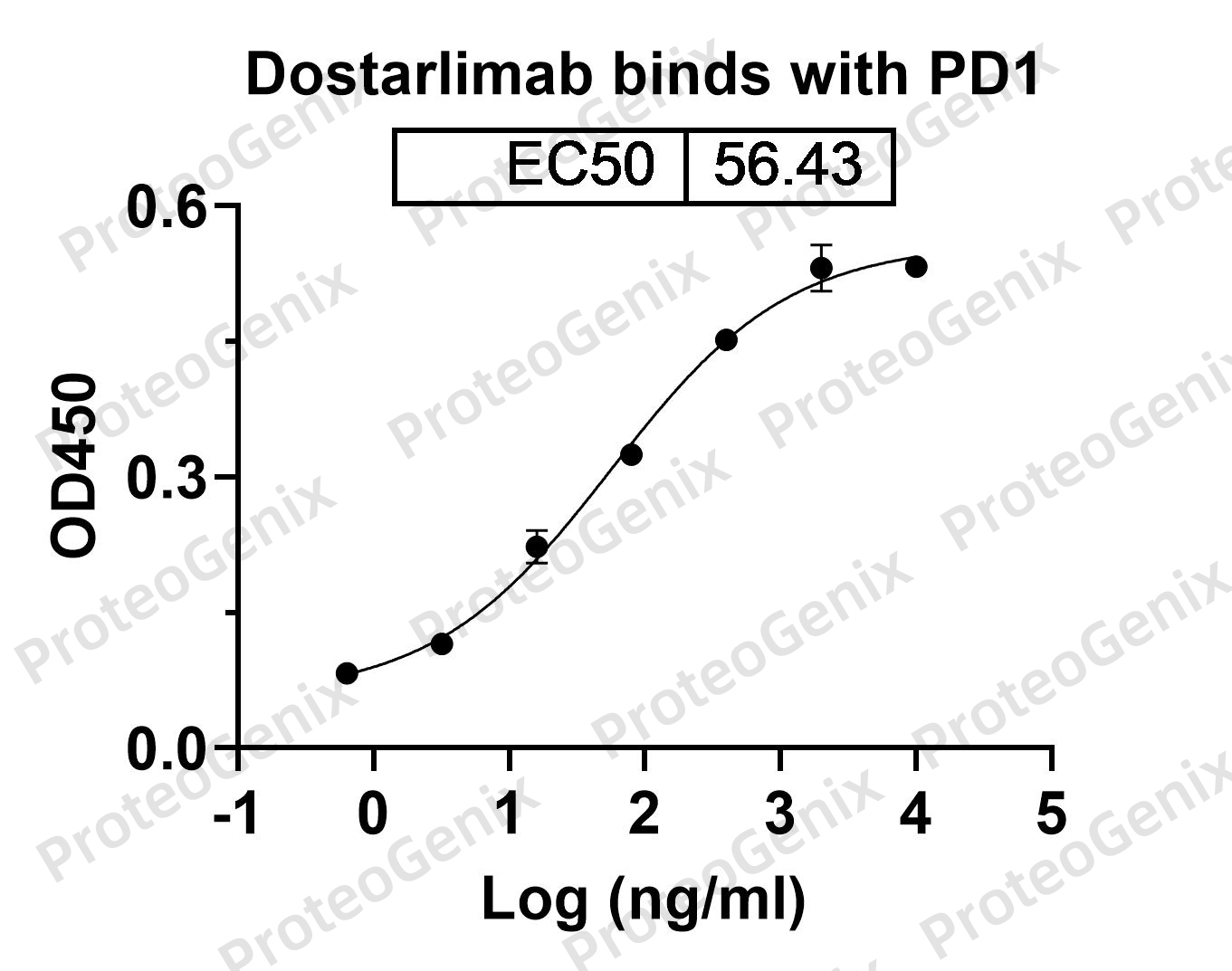
Dostarlimab biosimilar
Dostarlimab is a humanized monoclonal antibody. It aims to block programmed death receptor -1 (PD1). Dostarlimab has been approved in 2021 in the treatment of endometrial cancer under the brand name Jemperli.
About the target: PD1
Programmed death receptor-1, is also called Programmed cell death protein (PDCD1). It is the cluster of differenciation 279 (CD279). This protein is an immune checkpoint, it has an inhibbitory activity on immune response. Indeed it can be found at the surface of immune T-cell. Its neutralization is therefore involved in many therapeutic research areas.
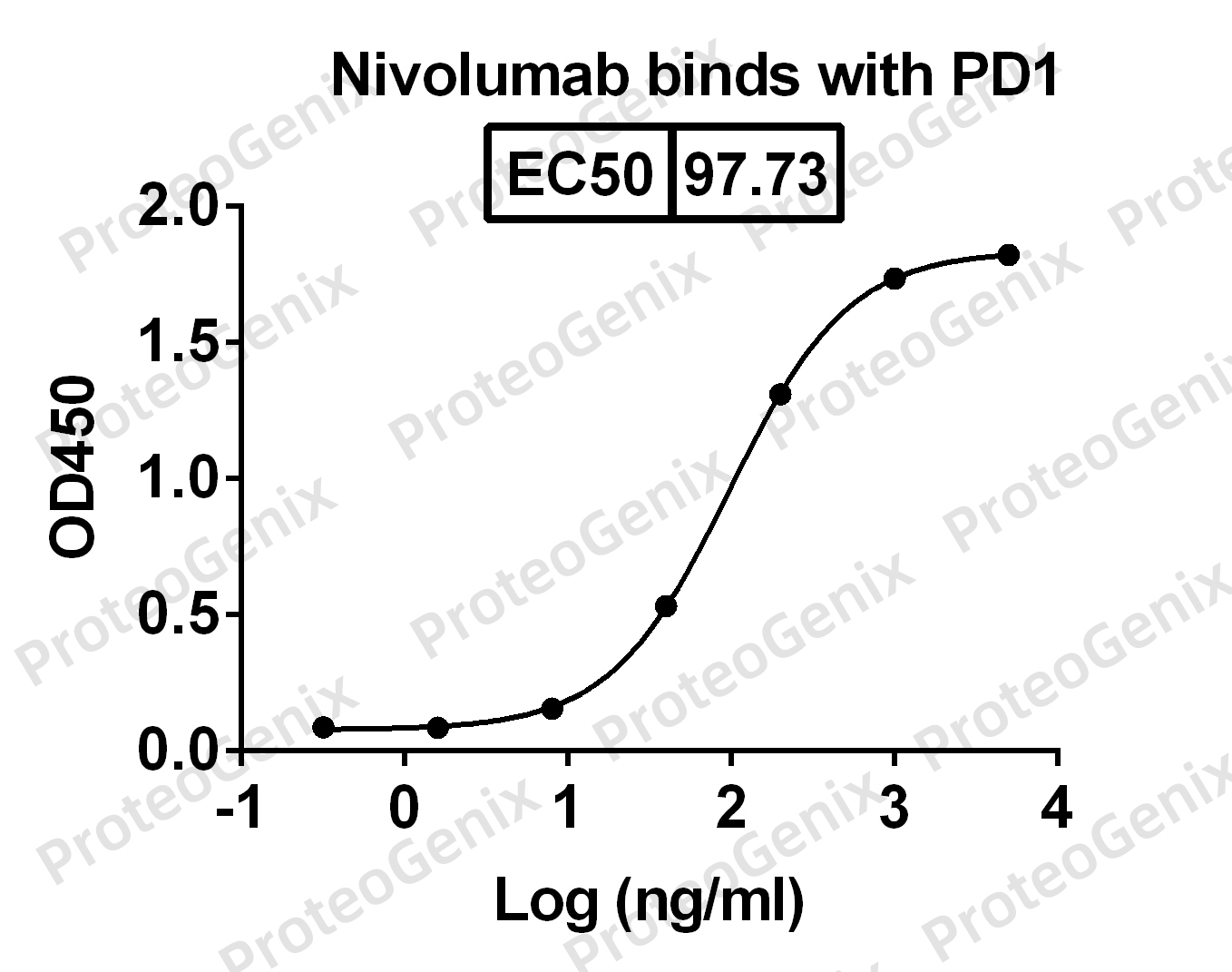
PD-1 has two ligands, PDL-1 and PDL-2. PDL1 are expressed at the surface of many cancer cells. Therefore they activate the negative response of PD1. The blockade of the interaction between PD1 and its ligand is thought to be a promissing treatment of cancers. In mouse models, PD-1 blockade, has been shown to improve CD8+ T cell function. Many monoclonal antibodies have been developped to target and neutralize the binding activity of PD1. In addition to Dostarlimab, Ipilimumab, Pembrolizumab and Nivolumab for example have shown good results.
Recent advances in Dostarlimab development
In early June 2022, a phase 2 study reported good results in the application of Dostarlimab in rectal cancer treatment. Indeed a cohort of 12 patients has received the treatment for 6 months. At the end of the period, cancer cells were found in none of them. Besides, no side effect of grade 3 or higher were reported. This study raised a major interest in the scientific community.
A research grade biosimilar
Dostarlimab biosimilar is a humanized antibody produced in ProteoGenix proprietary mammalian cell line XtenCHOTM (Chinese Hamster Ovary -CHO). Proteogenix is using a recombinant DNA technology. Proteogenix is offering this product for research use only. Dostarlimab biosimilar is not suitable for clinical or therapeutic use.

There are no reviews yet.