Rilotumumab Biosimilar – Anti-HGF mAb – Research Grade Rilotumumab Biosimilar – Anti-HGF mAb – Research Grade Introduction
Rilotumumab Biosimilar is a monoclonal antibody (mAb) that specifically targets hepatocyte growth factor (HGF). It is a research grade antibody that has shown promising results in pre-clinical studies and is currently being evaluated for its potential therapeutic applications.
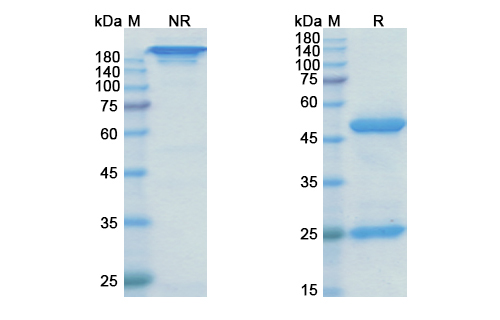
Structure of Rilotumumab Biosimilar
Rilotumumab Biosimilar is a humanized IgG1 monoclonal antibody, meaning it is derived from human antibodies and has been modified to reduce its immunogenicity. It has a molecular weight of approximately 150 kDa and consists of two heavy chains and two light chains. The heavy chains are made up of four constant domains (CH1, CH2, CH3, and CH4) and one variable domain (VH), while the light chains contain two constant domains (CL) and one variable domain (VL).
Rilotumumab Biosimilar binds to the alpha chain of the HGF receptor, c-Met, with high affinity and specificity. This binding inhibits the binding of HGF to c-Met, thereby preventing the activation of downstream signaling pathways involved in cell proliferation, survival, and migration.
Mechanism of Action
The primary function of Rilotumumab Biosimilar is to block the interaction between HGF and c-Met, which is a crucial pathway in the development and progression of many types of cancer. HGF is a growth factor that is overexpressed in various tumors and promotes tumor growth, invasion, and metastasis. By inhibiting the HGF/c-Met pathway, Rilotumumab Biosimilar can potentially slow down or stop the growth and spread of cancer cells.
Rilotumumab Biosimilar also has an indirect anti-tumor effect by stimulating antibody-dependent cell-mediated cytotoxicity (ADCC). This is a process where immune cells, such as natural killer (NK) cells, recognize and kill cancer cells that have been marked by Rilotumumab Biosimilar.
Research Applications
Rilotumumab Biosimilar is currently being evaluated in pre-clinical studies for its potential therapeutic applications in various types of cancer, including gastric, breast, lung, and colorectal cancer. It has shown promising results in inhibiting tumor growth and metastasis in animal models.
Furthermore, Rilotumumab Biosimilar has also been studied for its potential use in combination with other cancer treatments, such as chemotherapy and radiation therapy. Pre-clinical studies have shown that Rilotumumab Biosimilar can enhance the effectiveness of these treatments by sensitizing cancer cells to their effects.
Conclusion
Rilotumumab Biosimilar is a promising research grade antibody that specifically targets HGF and has the potential to be an effective treatment for various types of cancer. Its unique mechanism of action and potential for combination therapy make it a promising candidate for further clinical development. With ongoing research and clinical trials, Rilotumumab Biosimilar has the potential to improve the outcomes and quality of life for cancer patients.
There are no reviews yet.