Introduction
Sifalimumab Biosimilar, also known as Anti-IFNA1 mAb, is a novel monoclonal antibody that has been developed as a potential therapeutic agent for treating various autoimmune diseases. This biosimilar is a highly specific and potent inhibitor of interferon-alpha (IFN-α), a cytokine that plays a crucial role in the pathogenesis of autoimmune disorders. In this article, we will discuss the structure, activity, and potential applications of Sifalimumab Biosimilar in detail.
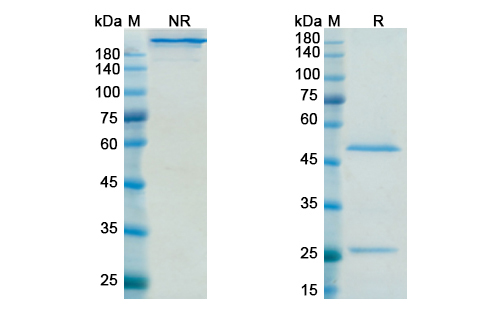
Structure of Sifalimumab Biosimilar
Sifalimumab Biosimilar is a recombinant humanized monoclonal antibody that has been designed to target and neutralize IFN-α. It is composed of two heavy chains and two light chains, with a total molecular weight of approximately 150 kDa. The antibody has a unique structure that allows it to bind specifically to the IFN-α receptor, blocking its signaling pathway and preventing the downstream effects of IFN-α. The amino acid sequence of Sifalimumab Biosimilar is highly homologous to the reference product, making it a highly similar and interchangeable biosimilar.
Activity of Sifalimumab Biosimilar
The primary mechanism of action of Sifalimumab Biosimilar is its ability to bind to IFN-α and inhibit its signaling. IFN-α is a type I interferon that plays a crucial role in the pathogenesis of autoimmune diseases, including systemic lupus erythematosus (SLE), dermatomyositis (DM), and Sjögren’s syndrome (SS). It is known to activate immune cells, promote inflammation, and contribute to tissue damage in these conditions. By blocking the activity of IFN-α, Sifalimumab Biosimilar can reduce the symptoms and disease progression of these autoimmune disorders.
In addition to its direct effects on IFN-α, Sifalimumab Biosimilar also has an immunomodulatory role. It has been shown to regulate the production of other cytokines and chemokines involved in the immune response, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α). This further contributes to its anti-inflammatory and disease-modifying effects.
Therapeutic Target of Sifalimumab Biosimilar
The primary therapeutic target of Sifalimumab Biosimilar is IFN-α, making it a promising treatment for autoimmune diseases where this cytokine is overproduced. Some of the specific indications for which Sifalimumab Biosimilar is being investigated include SLE, DM, and SS. These are chronic, debilitating conditions that can affect multiple organ systems and have a significant impact on patients’ quality of life. By targeting IFN-α, Sifalimumab Biosimilar has the potential to provide a more targeted and effective treatment option for these diseases.
Research Grade Sifalimumab Biosimilar
Sifalimumab Biosimilar is currently in the research stage, with several preclinical and clinical studies underway to evaluate its safety and efficacy. The biosimilar is being developed as a cost-effective alternative to the reference product, with the aim of providing more affordable treatment options for patients with autoimmune diseases. The research grade of Sifalimumab Biosimilar is being used in these studies to gather data on its pharmacokinetics, pharmacodynamics, and immunogenicity. Once the research is complete, the biosimilar will undergo regulatory review and may be approved for use in clinical practice.
Conclusion
Sifalimumab Biosimilar, also known as Anti-IFNA1 mAb, is a promising therapeutic agent for treating autoimmune diseases. Its unique structure and mechanism of action make it a highly specific and potent inhibitor of IFN-α, a key cytokine involved in the pathogenesis of these conditions. As a research grade biosimilar, it is currently
There are no reviews yet.