Tafolecimab Biosimilar: A Novel Anti-PCSK9 mAb for Therapeutic Targeting Tafolecimab Biosimilar: A Novel Anti-PCSK9 mAb for Therapeutic Targeting Introduction
Tafolecimab Biosimilar is a monoclonal antibody (mAb) that specifically targets proprotein convertase subtilisin/kexin type 9 (PCSK9), a protein involved in the regulation of low-density lipoprotein (LDL) cholesterol levels. This biosimilar is a highly promising therapeutic agent for the treatment of dyslipidemia and cardiovascular diseases.
Structure of Tafolecimab Biosimilar
Tafolecimab Biosimilar is a recombinant humanized IgG1 mAb, produced by using Chinese hamster ovary (CHO) cells. It is composed of two identical heavy chains and two identical light chains, each consisting of a variable region and a constant region. The variable region of the mAb is responsible for binding to PCSK9, while the constant region mediates effector functions.
Mechanism of Action
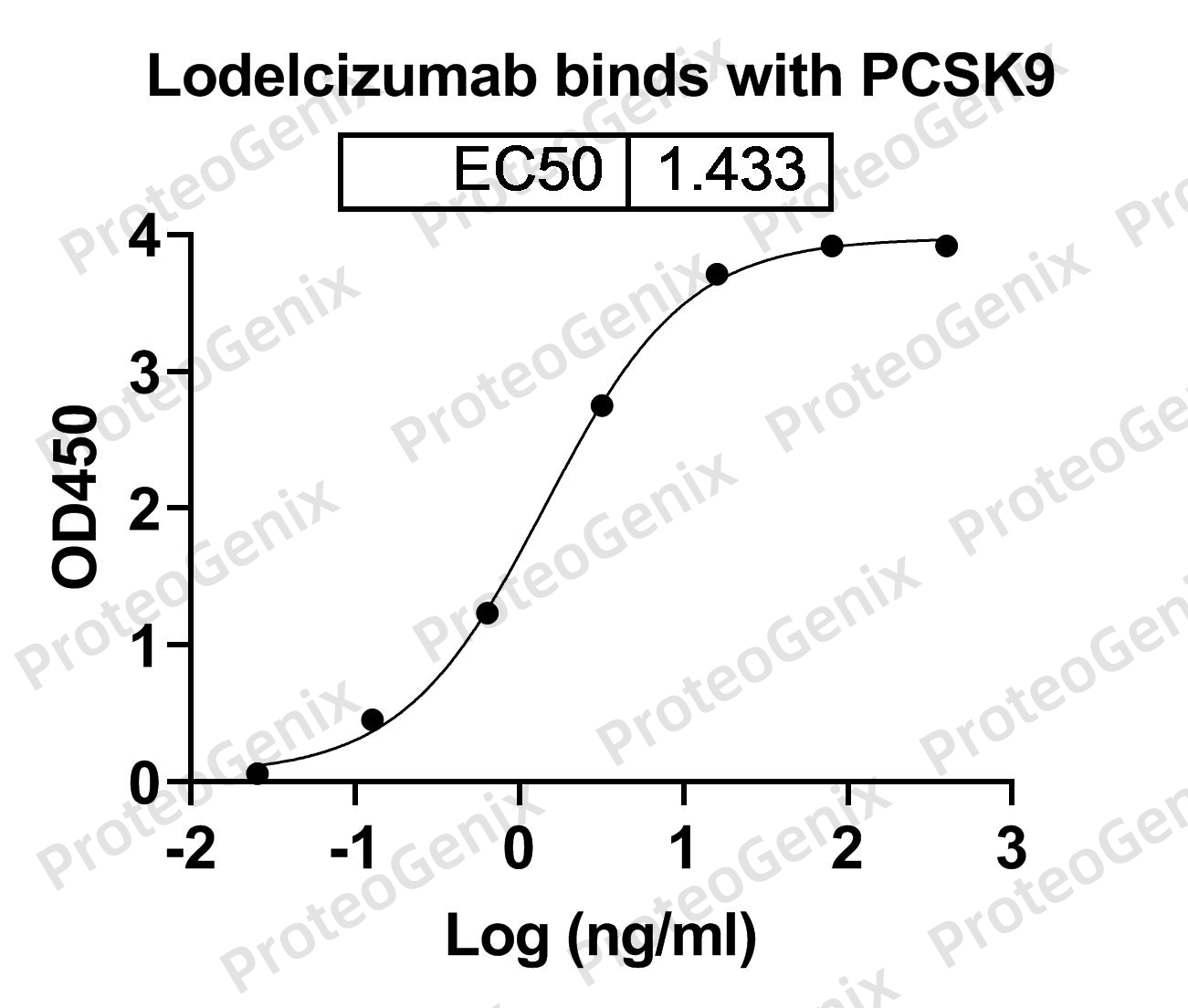
Tafolecimab Biosimilar binds to PCSK9 with high affinity and specificity, preventing it from binding to the LDL receptor (LDLR). PCSK9 normally binds to LDLR and promotes its degradation, leading to increased levels of LDL cholesterol in the blood. By blocking this interaction, Tafolecimab Biosimilar increases the number of LDLRs on the cell surface, resulting in increased uptake of LDL cholesterol from the blood and lowering of LDL cholesterol levels.
Therapeutic Applications
Tafolecimab Biosimilar has potential therapeutic applications in the treatment of dyslipidemia and cardiovascular diseases. It can be used as a monotherapy or in combination with statins or other lipid-lowering agents for the management of hypercholesterolemia. It has also shown promising results in reducing the risk of cardiovascular events in patients with a history of cardiovascular disease.
Advantages of Tafolecimab Biosimilar
Compared to other PCSK9 inhibitors, Tafolecimab Biosimilar has several advantages. It has a longer half-life, allowing for less frequent dosing, and has a lower risk of immunogenicity due to its humanized structure. It also has a lower production cost, making it more accessible for patients.
Clinical Trials
Tafolecimab Biosimilar has undergone several clinical trials to evaluate its safety and efficacy. In a phase III trial, it was shown to significantly reduce LDL cholesterol levels in patients with hypercholesterolemia. Another phase III trial demonstrated its ability to reduce the risk of cardiovascular events in patients with a history of cardiovascular disease. Ongoing trials are also evaluating its efficacy in combination with other lipid-lowering agents.
Conclusion
Tafolecimab Biosimilar is a promising therapeutic agent for the treatment of dyslipidemia and cardiovascular diseases. Its unique mechanism of action, favorable pharmacokinetic profile, and lower production cost make it a highly attractive option for patients. Ongoing clinical trials will further establish its efficacy and safety, paving the way for its approval and commercialization.
There are no reviews yet.