Tilogotamab Biosimilar – Anti-TNFRSF10B mAb – Research Grade Tilogotamab Biosimilar – Anti-TNFRSF10B mAb – Research Grade
Tilogotamab Biosimilar is a monoclonal antibody (mAb) that targets the tumor necrosis factor receptor superfamily member 10B (TNFRSF10B), also known as death receptor 5 (DR5). This biosimilar is a highly specific and potent therapeutic agent that has shown promising results in preclinical studies.
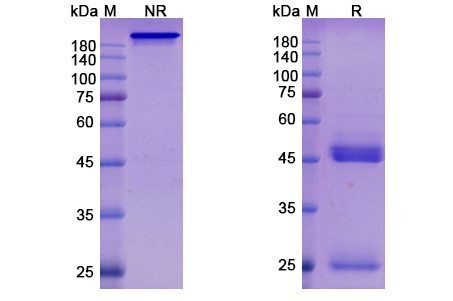
Structure of Tilogotamab Biosimilar
Tilogotamab Biosimilar is a fully humanized IgG1 antibody with a molecular weight of approximately 150 kDa. It is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region is responsible for binding to its target, TNFRSF10B, while the constant region is responsible for effector functions such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).
Mechanism of Action
Tilogotamab Biosimilar works by binding to TNFRSF10B on the surface of cancer cells. This binding triggers a signaling cascade that leads to the activation of caspases, which are enzymes responsible for inducing apoptosis (cell death). This mechanism is highly specific to cancer cells, sparing normal cells from damage.
Applications of Tilogotamab Biosimilar
Tilogotamab Biosimilar is being investigated for its potential use in the treatment of various types of cancer, including solid tumors and hematological malignancies. It has shown promising results in preclinical studies, demonstrating potent anti-tumor activity and minimal toxicity. Clinical trials are currently ongoing to evaluate its safety and efficacy in cancer patients.
Potential Advantages of Tilogotamab Biosimilar
As a biosimilar, Tilogotamab offers several potential advantages over its originator drug. These include lower production costs, increased accessibility for patients, and potentially improved efficacy and safety profiles. Additionally, Tilogotamab Biosimilar has the potential to be used in combination with other cancer therapies, such as chemotherapy and immune checkpoint inhibitors, to enhance its anti-tumor effects.
Conclusion
Tilogotamab Biosimilar is a promising therapeutic agent for the treatment of cancer. Its unique mechanism of action, high specificity, and potential advantages as a biosimilar make it a valuable addition to the arsenal of cancer treatments. Ongoing research and clinical trials will further elucidate its potential and pave the way for its use in the clinic.
Keywords
Antibody, therapeutic target, TNFRSF10B, Tilogotamab Biosimilar, monoclonal antibody, preclinical studies, cancer, apoptosis, caspases, biosimilar, originator drug, chemotherapy, immune checkpoint inhibitors, clinical trials.
There are no reviews yet.