PX-TA1005-MMAEd4-1MG is a research-grade antibody-drug conjugate (ADC) developed by ProteoGenix, based on a trastuzumab biosimilar (anti-HER2 monoclonal antibody). Designed for non-clinical use, it enables the study of ADC mechanisms and performance using a clinically validated antibody scaffold.
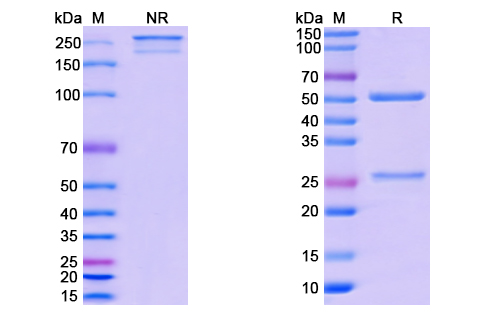
This antibody is conjugated to Monomethyl Auristatin E (MMAE)—a highly potent antimitotic agent—via a cathepsin-cleavable c-ValCitPAB linker. This well-characterized linker-payload system ensures that the drug is released selectively within the target cell’s lysosomal environment. The conjugate has a Drug-to-Antibody Ratio (DAR) of 4, allowing optimal balance between efficacy and stability.
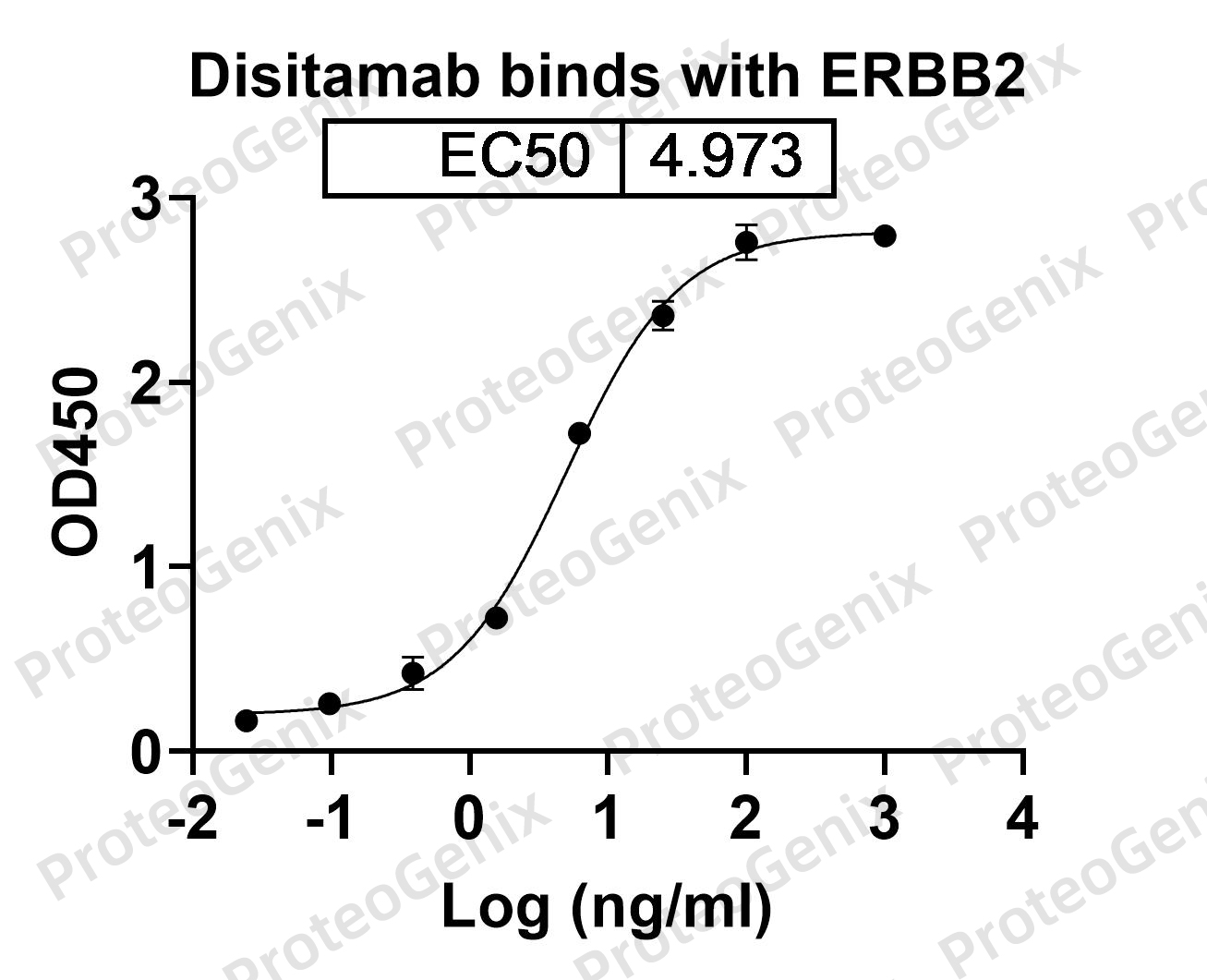
PX-TA1005-MMAEd4-1MG retains the HER2-binding properties of trastuzumab, enabling target-specific delivery of MMAE to HER2-positive cells. It is particularly well-suited for:
- Evaluating ADC cytotoxicity in HER2-overexpressing tumor models
- Studying internalization and trafficking of HER2-targeted payloads
- Benchmarking experimental ADCs against a known antibody framework
- Conducting biodistribution and pharmacokinetic studies in preclinical models
This product is ideal for academic and industrial teams involved in oncology drug discovery, ADC platform validation, or payload optimization. As a trastuzumab biosimilar, it offers full compatibility with established HER2 research tools and models, while eliminating the cost and regulatory constraints of therapeutic-grade antibodies.
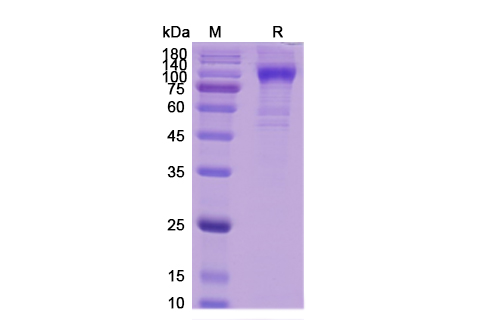
Manufactured to high research standards, PX-TA1005-MMAEd4-1MG ensures reproducibility and consistency for robust experimental results in ADC development pipelines.
There are no reviews yet.