Introduction
Tulinercept Biosimilar is a novel anti-TNF fusion protein that has been developed as a potential therapeutic for various autoimmune and inflammatory diseases. This biosimilar is a research grade product that is designed to mimic the structure and function of an existing anti-TNF antibody, while offering improved efficacy and safety profiles.
Structure of Tulinercept Biosimilar
Tulinercept Biosimilar is a fusion protein that consists of two distinct domains – a human TNF receptor extracellular domain and a human IgG1 Fc fragment. The TNF receptor domain is responsible for binding to TNF, while the Fc fragment provides the effector functions of the antibody, such as complement activation and antibody-dependent cellular cytotoxicity.
The TNF receptor domain of Tulinercept Biosimilar is a soluble form of the human TNF receptor 1 (TNFR1), also known as p55 or CD120a. This domain contains the cysteine-rich domain 1 (CRD1) and CRD2, which are responsible for binding to TNF. The Fc fragment of the antibody is derived from the constant region of a human IgG1 antibody and contains the hinge, CH2, and CH3 domains.
Mechanism of Action
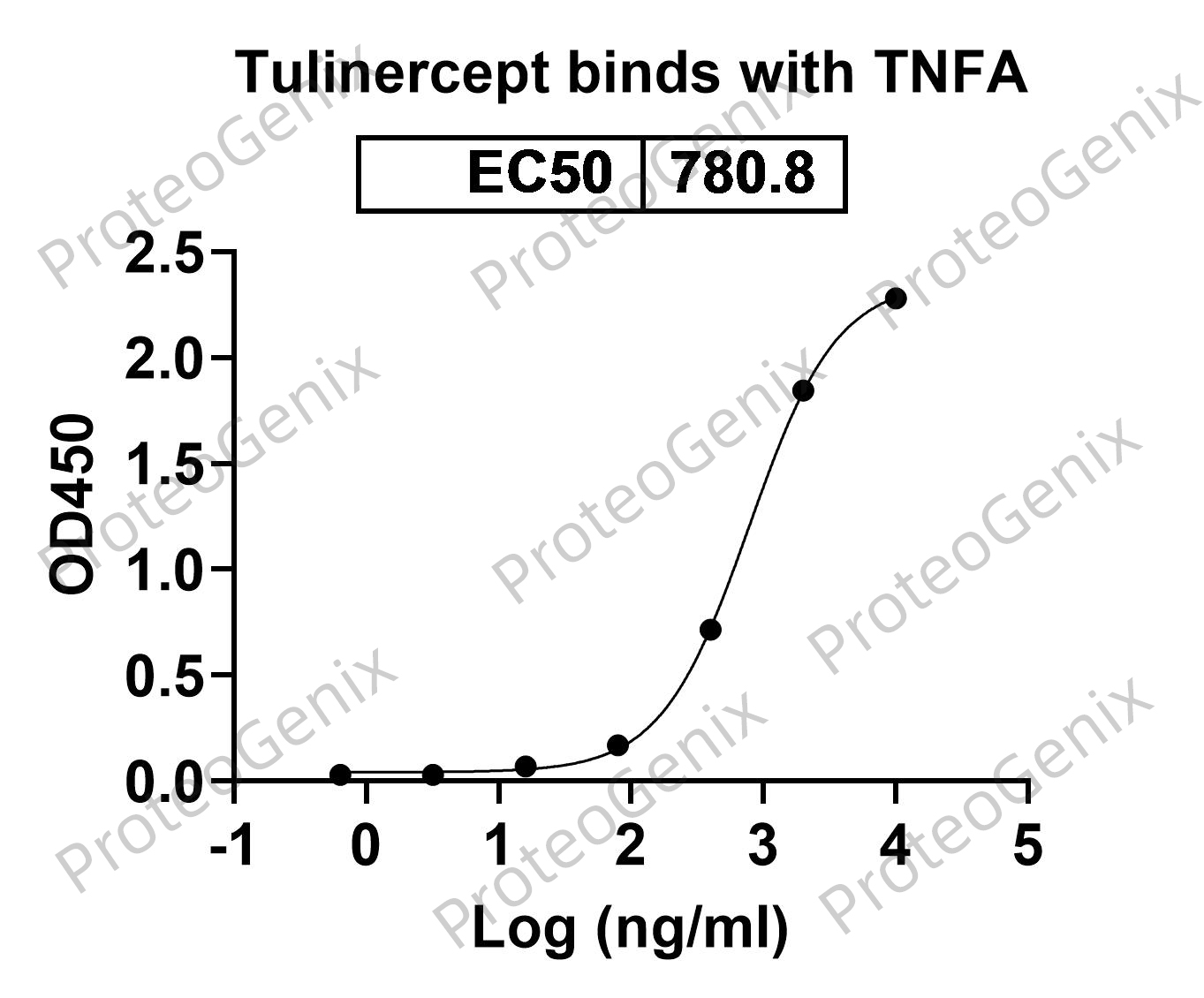
Tulinercept Biosimilar acts as a competitive inhibitor of TNF by binding to TNF and preventing its interaction with its receptors on the cell surface. This leads to a decrease in the downstream signaling pathways that are activated by TNF, thereby reducing the inflammatory response.
In addition, the Fc fragment of Tulinercept Biosimilar can also activate complement, leading to the destruction of cells that express TNF on their surface. This mechanism is particularly useful in diseases where TNF-producing cells are present, such as rheumatoid arthritis.
Therapeutic Targets
The primary therapeutic target of Tulinercept Biosimilar is TNF, a pro-inflammatory cytokine that plays a crucial role in the pathogenesis of various autoimmune and inflammatory diseases. TNF is produced by multiple cell types, including macrophages, T cells, and fibroblasts, and is involved in the regulation of immune responses and inflammation.
Tulinercept Biosimilar has shown promising results in clinical trials for the treatment of diseases such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease. These diseases are characterized by high levels of TNF, and the inhibition of TNF by Tulinercept Biosimilar has been shown to improve symptoms and reduce disease activity.
Applications of Tulinercept Biosimilar
Tulinercept Biosimilar has the potential to be used as a first-line treatment for various autoimmune and inflammatory diseases, either as a monotherapy or in combination with other therapies. Its unique structure and mechanism of action make it a promising candidate for patients who have not responded to other anti-TNF therapies.
Furthermore, Tulinercept Biosimilar can also be used in research settings to study the role of TNF in various diseases and to develop new treatments targeting TNF. Its research grade status allows for easier access and use in laboratory studies.
Conclusion
In summary, Tulinercept Biosimilar is a novel anti-TNF fusion protein with a unique structure and mechanism of action. It acts as a competitive inhibitor of TNF and also has the ability to activate complement, making it a promising therapeutic for various autoimmune and inflammatory diseases. Its potential applications in both clinical and research settings make it a valuable addition to the arsenal of anti-TNF therapies.
There are no reviews yet.