Introduction
Vudalimab Biosimilar, also known as Anti-CTLA4, PDCD1 mAb, is a research grade antibody that has shown promising results in the field of immunotherapy. This biosimilar is designed to target two important immune checkpoints, CTLA4 and PDCD1, which play a crucial role in regulating the immune response. In this article, we will delve into the structure, activity, and application of Vudalimab Biosimilar in the field of cancer research.
Structure of Vudalimab Biosimilar
Vudalimab Biosimilar is a monoclonal antibody that is produced by recombinant DNA technology. It is a fully humanized antibody, meaning that it is derived from human genes and has a structure similar to the antibodies naturally produced in the human body. This makes it less likely to cause an immune response when administered to patients.
The antibody has a Y-shaped structure, with two identical heavy chains and two identical light chains. Each chain consists of variable and constant regions. The variable regions are responsible for binding to the target molecules, CTLA4 and PDCD1, while the constant regions determine the antibody’s effector functions.
Activity of Vudalimab Biosimilar
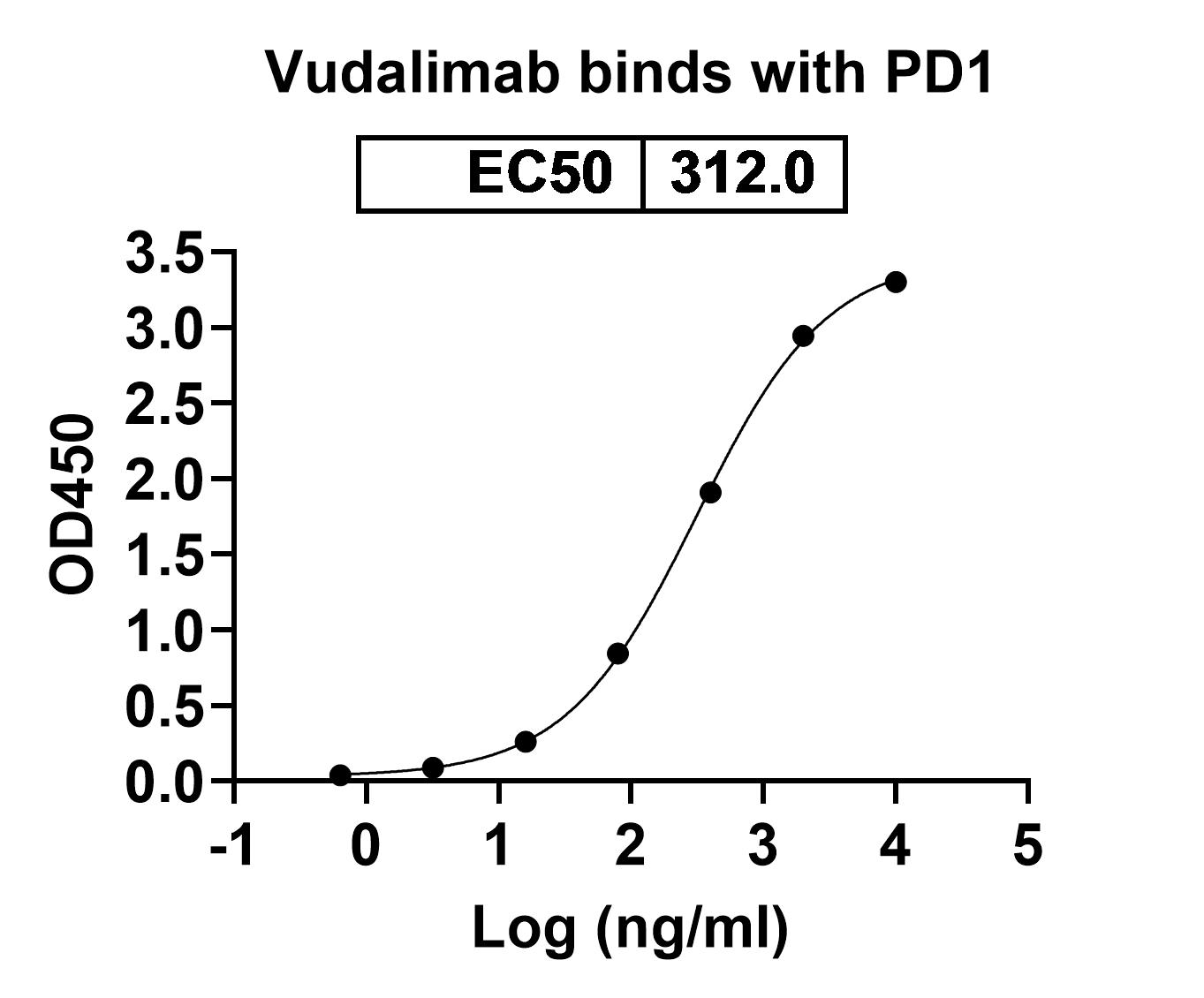
Vudalimab Biosimilar is a potent inhibitor of CTLA4 and PDCD1, which are immune checkpoints that are overexpressed in many types of cancer. These checkpoints play a crucial role in regulating the immune response by inhibiting T-cell activation. However, in cancer, these checkpoints are upregulated, leading to the suppression of the immune response and allowing cancer cells to escape detection and destruction by the immune system.
By targeting CTLA4 and PDCD1, Vudalimab Biosimilar blocks their inhibitory effects, allowing T-cells to become activated and mount an effective immune response against cancer cells. This activity has been shown to be effective in various preclinical and clinical studies, making Vudalimab Biosimilar a promising candidate for cancer immunotherapy.
Application of Vudalimab Biosimilar
Vudalimab Biosimilar is primarily being studied for its potential use in the treatment of various types of cancer, including melanoma, lung cancer, and bladder cancer. It is being evaluated as both a monotherapy and in combination with other cancer treatments, such as chemotherapy and other immunotherapies.
In preclinical studies, Vudalimab Biosimilar has shown promising results in inhibiting tumor growth and improving survival rates in animal models. In clinical trials, it has shown good safety and tolerability profiles, with some patients showing positive responses to treatment.
Apart from
cancer, Vudalimab Biosimilar is also being studied for its potential use in other diseases, such as autoimmune disorders, where immune checkpoint inhibitors have shown promising results. It is also being explored for its use in combination with other immunotherapies to enhance their efficacy.
Conclusion
In conclusion, Vudalimab Biosimilar is a research grade antibody that targets the immune checkpoints CTLA4 and PDCD1. Its structure, activity, and application make it a promising candidate for cancer immunotherapy. As research and clinical trials continue, it is hoped that Vudalimab Biosimilar will become a valuable addition to the arsenal of treatments available for cancer and other diseases.
There are no reviews yet.