Introduction to Cibisatamab Biosimilar – A Novel Antibody Targeting CEACAM5&CD3E for
Cancer Treatment
Cibisatamab Biosimilar, also known as Anti-CEACAM5&CD3E,CD3E mAb – Research Grade, is a novel antibody that has shown promising results in cancer treatment. This biosimilar is designed to target two specific proteins, CEACAM5 and CD3E, which are known to play a crucial role in cancer development and progression. In this article, we will provide a detailed scientific description of the structure, activity, and potential applications of Cibisatamab Biosimilar.
Structure of Cibisatamab Biosimilar
Cibisatamab Biosimilar is a monoclonal antibody, which means it is derived from a single type of immune cell. It is a chimeric antibody, meaning it is composed of both human and mouse parts. The antibody is composed of two main regions – the Fab region and the Fc region. The Fab region is responsible for binding to the target proteins, while the Fc region is involved in immune system activation.
Activity of Cibisatamab Biosimilar
Cibisatamab Biosimilar is designed to target two proteins, CEACAM5 and CD3E. CEACAM5, also known as carcinoembryonic antigen-related cell adhesion molecule 5, is a cell surface protein that is overexpressed in many types of cancer, including colorectal, lung, and breast cancer. CD3E, on the other hand, is a protein found on the surface of T cells, a type of immune cell that plays a crucial role in fighting cancer.
When Cibisatamab Biosimilar binds to CEACAM5, it blocks the function of this protein, which is known to promote tumor growth and spread. At the same time, the antibody binds to CD3E, activating T cells and enhancing their ability to kill cancer cells. This dual mechanism of action makes Cibisatamab Biosimilar a potent anti- cancer agent.
Applications of Cibisatamab Biosimilar
Cibisatamab Biosimilar is currently being studied as a potential treatment for various types of cancer, including colorectal, lung, and breast cancer. It has shown promising results in preclinical studies and is now being evaluated in clinical trials. In a phase I clinical trial, Cibisatamab Biosimilar has shown promising anti-tumor activity in patients with advanced colorectal cancer. It has also been shown to have a favorable safety profile, with no serious adverse events reported.
In addition to its potential as a standalone therapy, Cibisatamab Biosimilar is also being studied in combination with other anti- cancer agents. For example, it is being evaluated in combination with chemotherapy in patients with advanced colorectal cancer. This combination therapy has shown promising results in preclinical studies and is now being evaluated in clinical trials.
Conclusion
Cibisatamab Biosimilar is a novel antibody that has shown promising results in cancer treatment. Its unique structure and dual mechanism of action make it a potent anti- cancer agent. It is currently being evaluated in clinical trials for various types of cancer and has shown promising results in preclinical studies. With further research and development, Cibisatamab Biosimilar has the potential to become a valuable addition to the arsenal of anti- cancer therapies.
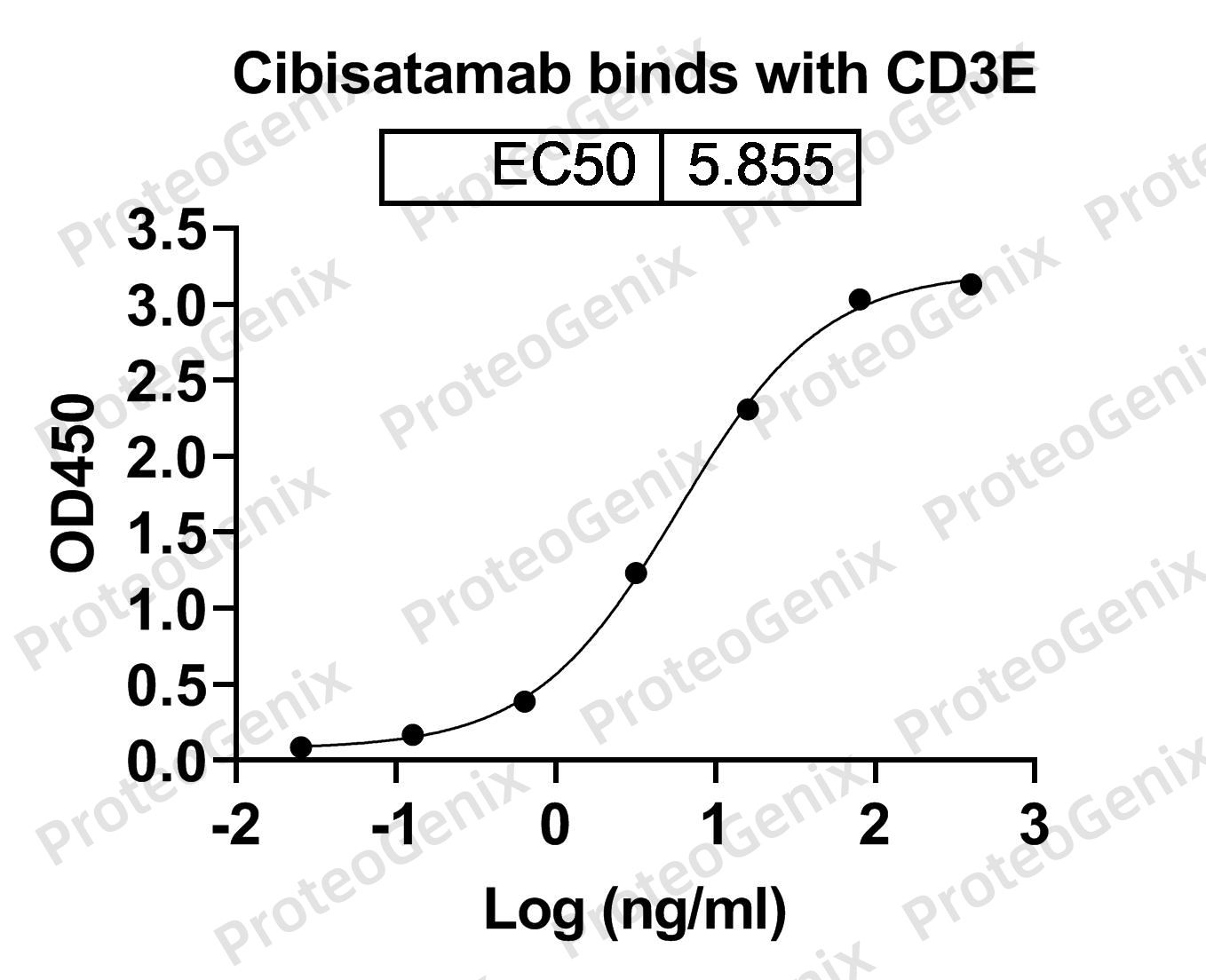
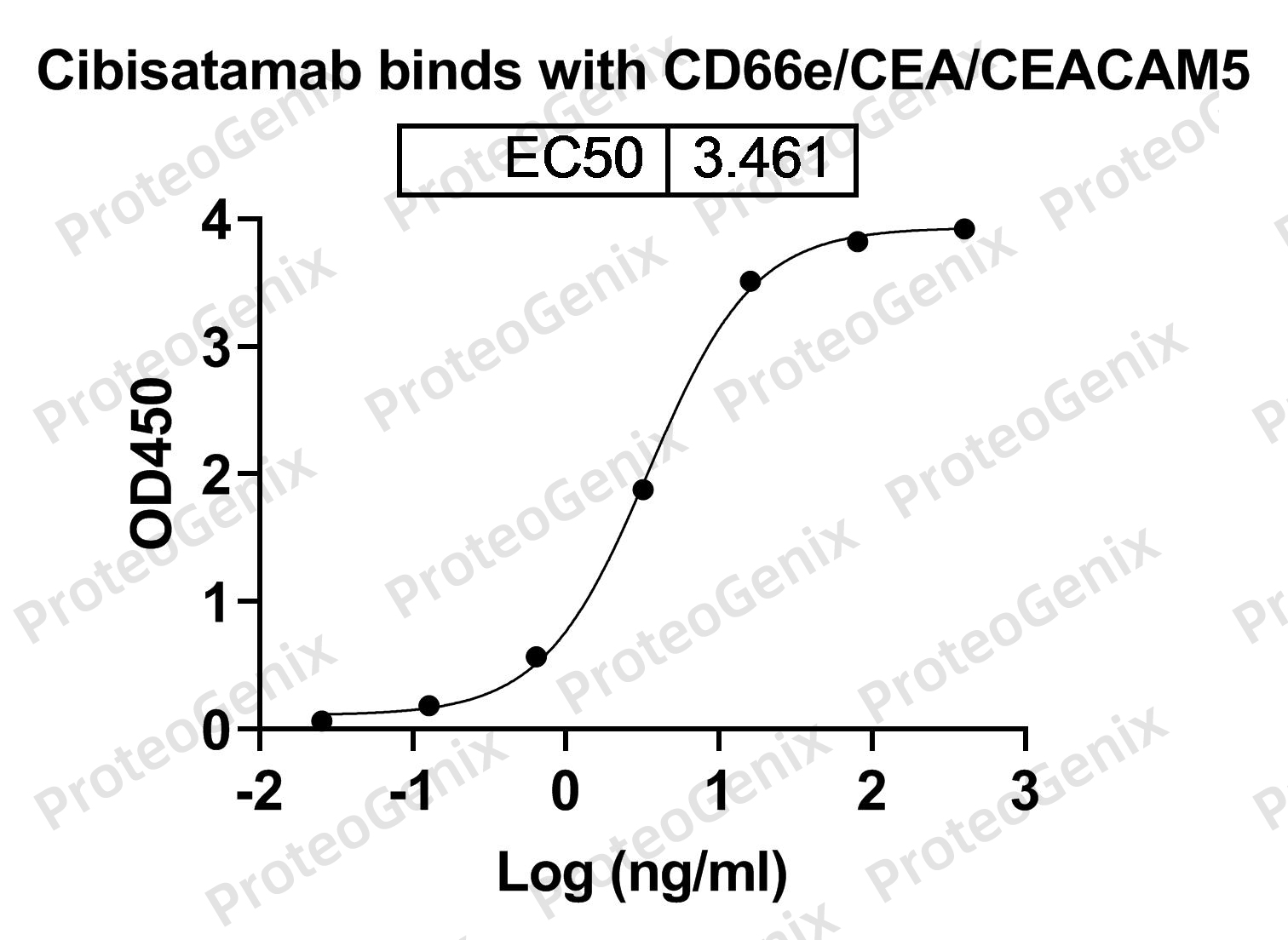
There are no reviews yet.