Introduction
Gevokizumab Biosimilar, also known as Anti-IL1B mAb, is a therapeutic antibody that targets the inflammatory cytokine interleukin-1 beta (IL-1B). This biosimilar is a research grade version of the original gevokizumab, which was developed by XOMA Corporation and is currently in clinical trials for various inflammatory diseases. In this article, we will explore the structure, activity, and potential applications of Gevokizumab Biosimilar in more detail.
Structure of Gevokizumab Biosimilar
Gevokizumab Biosimilar is a monoclonal antibody (mAb) that is produced from a single type of immune cell. It is a humanized antibody, meaning that it is derived from non-human sources but has been genetically modified to be more similar to human antibodies. The structure of gevokizumab biosimilar consists of two heavy chains and two light chains, which are linked together by disulfide bonds. The variable regions of the antibody, which are responsible for binding to IL-1B, are located at the tips of the heavy and light chains.
Activity of Gevokizumab Biosimilar
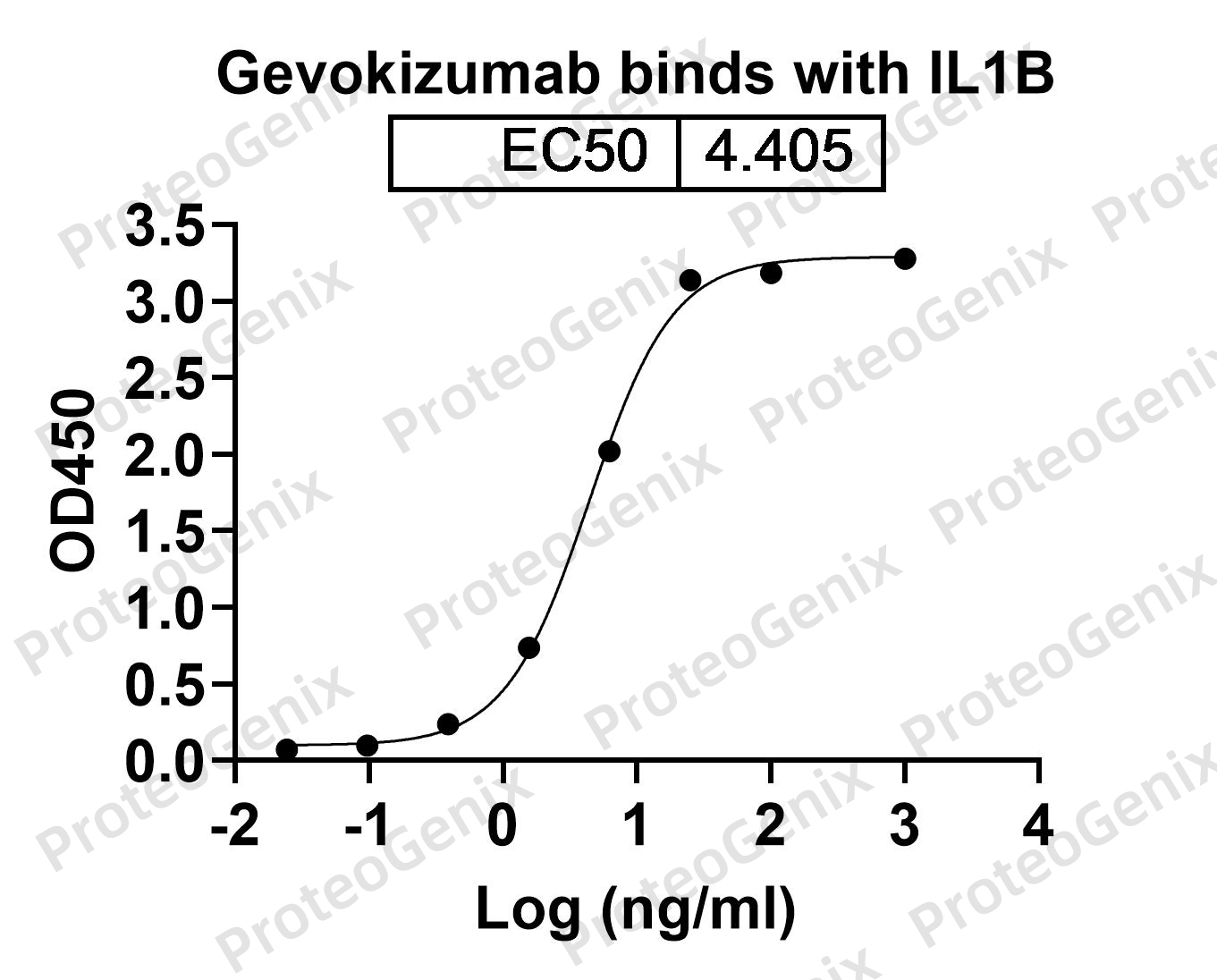
The main function of Gevokizumab Biosimilar is to bind to and neutralize IL-1B. IL-1B is a pro-inflammatory cytokine that plays a key role in the body’s immune response. It is produced by various cells, including macrophages and monocytes, and can cause inflammation and tissue damage when present in high levels. By binding to IL-1B, gevokizumab biosimilar prevents it from interacting with its receptors on target cells, thereby reducing its pro-inflammatory effects.
Applications of Gevokizumab Biosimilar
Gevokizumab Biosimilar has potential applications in various inflammatory diseases, as IL-1B is involved in the pathogenesis of many of these conditions. One of the main therapeutic targets for gevokizumab biosimilar is rheumatoid arthritis (RA), a chronic autoimmune disease characterized by joint inflammation and damage. In a phase II clinical trial, gevokizumab was shown to significantly reduce disease activity and improve symptoms in patients with RA.
In addition to RA, gevokizumab biosimilar may also be beneficial in other inflammatory conditions such as gout, osteoarthritis, and psoriasis. It has also shown promise in the treatment of non-infectious uveitis, a form of eye inflammation. In a phase III clinical trial, gevokizumab was found to be effective in reducing the recurrence of uveitis in patients with Behçet’s disease.
Future Directions
As gevokizumab biosimilar is still in the early stages of development, there is much potential for further research and clinical trials to explore its use in other inflammatory diseases. One area of interest is in the treatment of cytokine storm syndrome, a severe immune response that can occur in patients with COVID-19 and other viral infections. IL-1B is thought to play a role in this syndrome, and gevokizumab biosimilar may be a potential treatment option.
Conclusion
In summary, Gevokizumab Biosimilar is a research grade anti-IL1B mAb with a similar structure and activity to the original gevokizumab. It has the potential to be an effective treatment for various inflammatory diseases by targeting IL-1B, a key pro-inflammatory cytokine. With ongoing research and clinical trials, gevokizumab biosimilar may become a valuable therapeutic option for patients with these conditions.

There are no reviews yet.