Introduction to Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb
Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb is a monoclonal antibody (mAb) that has been developed as a biosimilar to the original Pateclizumab, which is a humanized mAb. This biosimilar has been designed to target lymphotoxin-alpha (LTA) and tumor necrosis factor superfamily member 1 (TNFSF1), both of which are important therapeutic targets in various diseases. In this article, we will explore the structure, activity, and applications of Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb.
Structure of Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb
Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb is a recombinant, fully humanized IgG1 mAb with a molecular weight of approximately 150 kDa. It is composed of two heavy chains and two light chains, each consisting of constant and variable regions. The variable regions of the antibody are responsible for binding to the target molecules, LTA and TNFSF1.
Activity of Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb
Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb exerts its activity by binding to LTA and TNFSF1, which are both pro-inflammatory cytokines involved in various disease processes. LTA is a potent inducer of inflammation and is known to play a key role in the development of autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus. TNFSF1 is also involved in inflammation and has been linked to diseases such as psoriasis and inflammatory bowel disease.
By binding to LTA and TNFSF1, Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb blocks their activity and reduces inflammation. This can lead to the improvement of symptoms and disease progression in patients with these conditions.
Applications of Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb
Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb has potential applications in various diseases where LTA and TNFSF1 play a role. It is currently being studied in clinical trials for the treatment of rheumatoid arthritis, systemic lupus erythematosus, psoriasis, and inflammatory bowel disease.
In rheumatoid arthritis, Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb has shown promising results in reducing disease activity and improving symptoms in patients who have not responded well to other treatments. It has also shown potential in slowing down joint damage and improving physical function.
In systemic lupus erythematosus, Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb has shown potential in reducing disease activity and improving organ involvement. It has also been found to be well-tolerated in patients with this condition.
In psoriasis, Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb has shown promising results in reducing skin lesions and improving quality of life in patients who have not responded well to other treatments.
In inflammatory bowel disease, Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb has shown potential in reducing disease activity and improving symptoms in patients with moderate to severe Crohn’s disease.
Conclusion
Pateclizumab Biosimilar – Anti-LTA, TNFSF1 mAb is a promising therapeutic option for various diseases where LTA and TNFSF1 play a role. Its structure, activity, and potential applications make it a valuable addition to the treatment options available for patients with conditions such as rhe
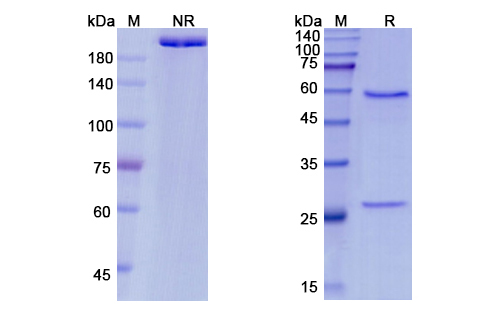
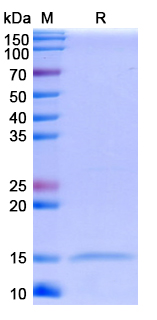
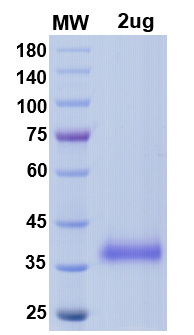
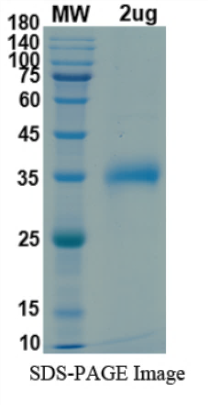
There are no reviews yet.