Cart (0 Items)
Your cart is currently empty.
View Products$238.00
100ug 0% OFF + 238 loyalty points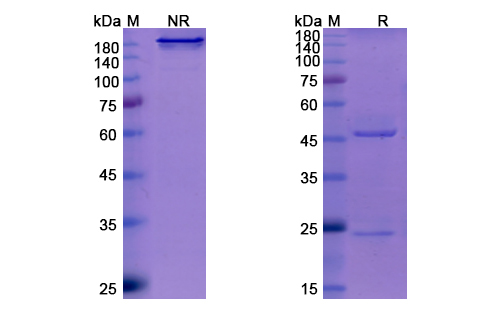
Sotrovimab Biosimilar - Anti-Spike glycoprotein mAb, on SDS-PAGE under reducing and non-reducing conditions. The gel was stained overnight with Coomassie Blue. The purity of the antibody is greater than 95%.
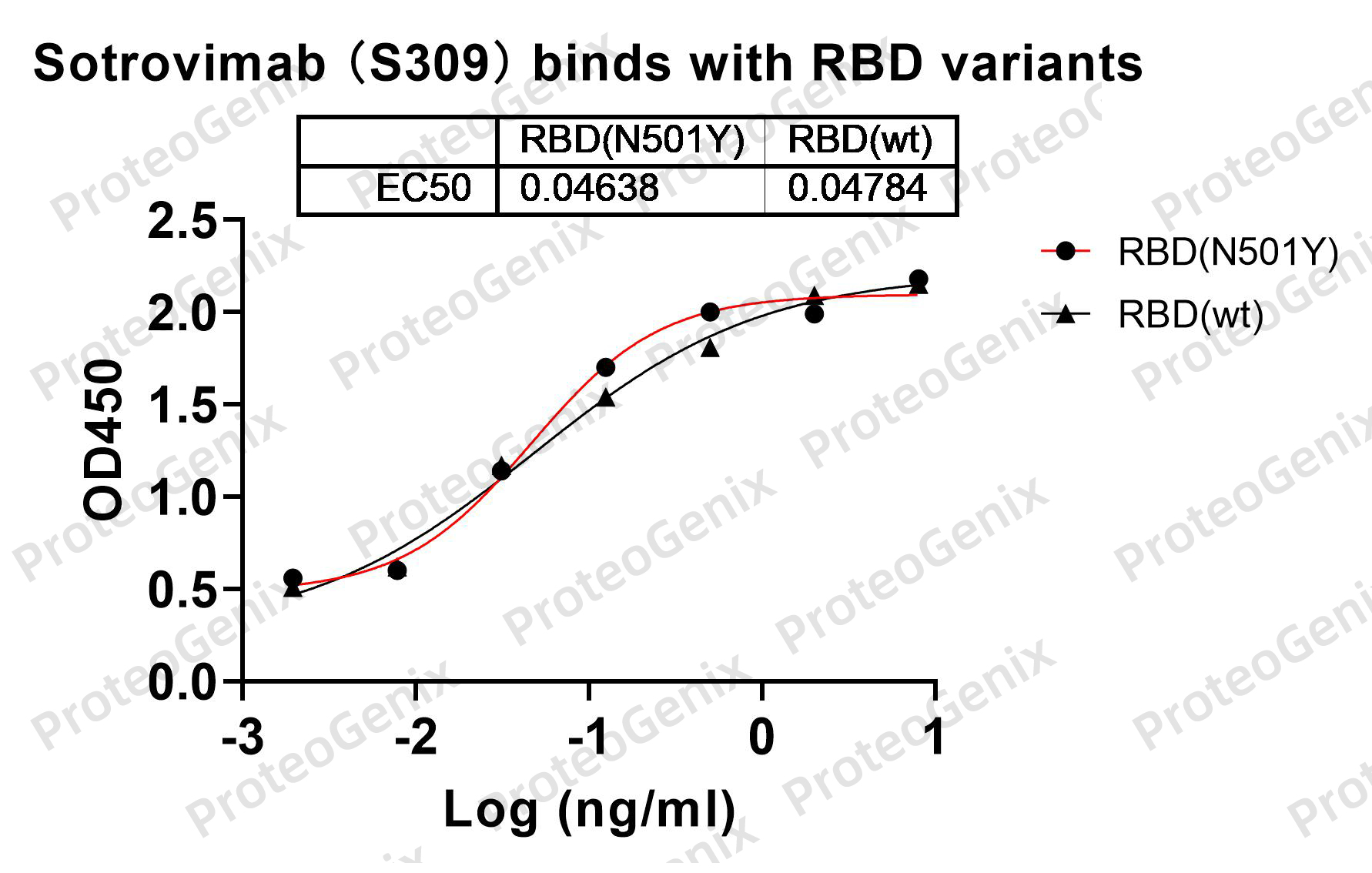
Immobilized RBD Domain (cat. No.PX-COV-P046) at 0.5µg/mL (100µL/well) can bind to Sotrovimab Biosimilar - Anti-Spike glycoprotein mAb (cat. No.PX-TA1637) in indirect ELISA with Goat Anti-Human IgG secondary antibody coupled with HRP measured by OD450
![SDS-PAGE for CoV-S[679-833] Recombinant proteins](https://www.proteogenix.science/wp-content/uploads/documents/validation/sds-validation-cov-s679-833.png)
PX-COV-P011-10
![SDS-PAGE for CoV-S [819-919] Recombinant proteins](https://www.proteogenix.science/wp-content/uploads/documents/validation/sds-validation-cov-s-819-919.png)
PX-COV-P014-10
PX-COV-P002-10
Our catalog doesn’t cover everything — but our team does. Whether you need a custom antibody, a specific protein variant, or a bulk order, our scientists are here to help.
Toshi — October 3, 2021
★★★☆☆
This antibody along with other therapeutic antibodies were tested in surrogate virus neutralization test using spike trimers and ACE2 protein. This antibody did not show weak inhibition of all variants tested (IC50 344.03 ng/mL for Delta, >500 ng/mL for Gamma and Alpha, 244.1 ng/mL for the wild type).