Teplizumab Biosimilar – Anti-CD3 mAb – Research Grade
Teplizumab Biosimilar – Anti-CD3 mAb – Research Grade: A Promising Antibody for Targeted Therapy
Introduction
Teplizumab Biosimilar, also known as Anti-CD3 monoclonal antibody (mAb), is a novel therapeutic antibody that has shown promising results in the treatment of various autoimmune diseases. It is a biosimilar version of the FDA-approved drug Teplizumab, which is used for the treatment of type 1 diabetes. This research grade antibody has been extensively studied for its structure, activity, and potential applications in the field of targeted therapy.
Structure of Teplizumab Biosimilar
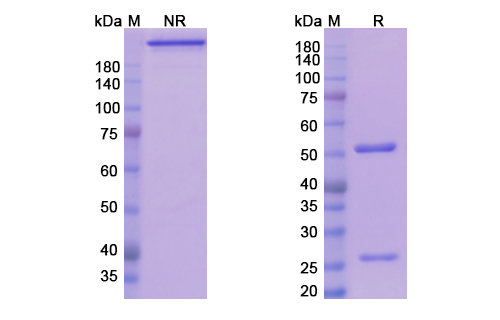
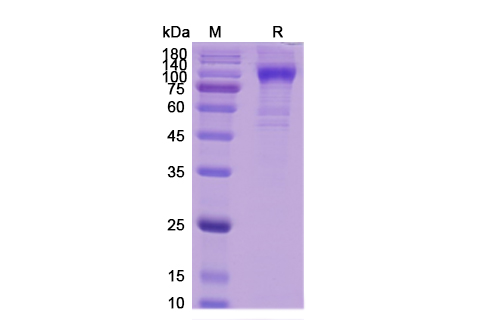
Teplizumab Biosimilar is a humanized IgG1 monoclonal antibody that specifically targets the CD3 antigen on T-cells. It is composed of two heavy chains and two light chains, each containing a variable region and a constant region. The variable region of the antibody is responsible for binding to the CD3 antigen, while the constant region mediates effector functions such as complement-dependent cytotoxicity and antibody-dependent cellular cytotoxicity.
The structure of Teplizumab Biosimilar is highly similar to that of the original Teplizumab, with minor differences in the amino acid sequence. This biosimilar version has been carefully engineered to maintain the same binding affinity and specificity as the original drug, while also ensuring a high level of purity and stability. This makes it a reliable and effective therapeutic option for targeted therapy.
Activity of Teplizumab Biosimilar
Teplizumab Biosimilar works by binding to the CD3 antigen on T-cells, which are a key component of the immune system. This binding leads to the activation of T-cells and subsequent suppression of immune response. In autoimmune diseases, the immune system mistakenly attacks healthy cells and tissues, causing inflammation and damage. By targeting and inhibiting the activity of T-cells, Teplizumab Biosimilar helps to reduce this harmful immune response and alleviate symptoms of autoimmune diseases.
Studies have shown that Teplizumab Biosimilar has a high affinity for the CD3 antigen, allowing it to effectively block T-cell activation. It has also been found to have a long half-life, which means it can remain in the body for an extended period of time, providing sustained therapeutic effects.
Applications of Teplizumab Biosimilar
Teplizumab Biosimilar has been primarily studied for its potential applications in the treatment of autoimmune diseases such as type 1 diabetes, multiple sclerosis, and rheumatoid arthritis. In clinical trials, it has shown promising results in reducing disease activity and improving symptoms in patients with these conditions.
Additionally, Teplizumab Biosimilar has also been investigated for its potential use in organ transplantation. By suppressing T-cell activity, it can help prevent organ rejection and improve transplant outcomes. Furthermore, this antibody has also shown potential in the treatment of certain types of cancer, as it can induce apoptosis (cell death) in cancer cells.
Conclusion
Teplizumab Biosimilar, a biosimilar version of the FDA-approved drug Teplizumab, is a promising antibody for targeted therapy. Its highly similar structure and activity to the original drug, along with its potential applications in various autoimmune diseases and transplantation, make it a valuable addition to the field of medicine. Further research and clinical trials are needed to fully understand the potential of this biosimilar in improving the lives of patients with autoimmune diseases and other conditions.
There are no reviews yet.