Cart (0 Items)
Your cart is currently empty.
View ProductsIon Exchange Chromatography (IEX) is a protein purification technique that separates proteins based on their different electrostatic interactions with charged ligands at a specific pH. The latter pH is determined based on the pH-Isoelectric Point (pI) at which the net electrical charge of the protein is equal to zero. Therefore, the pH buffer should either be higher or inferior to the pI of the protein of interest.
The following section introduces the basic principles of Ion exchange chromatography and helps you understand when to choose this technique for your protein expression strategy.
Please note that this service is included in our protein expression packages if relevant.

Contact us to benefit from our full protein expression services
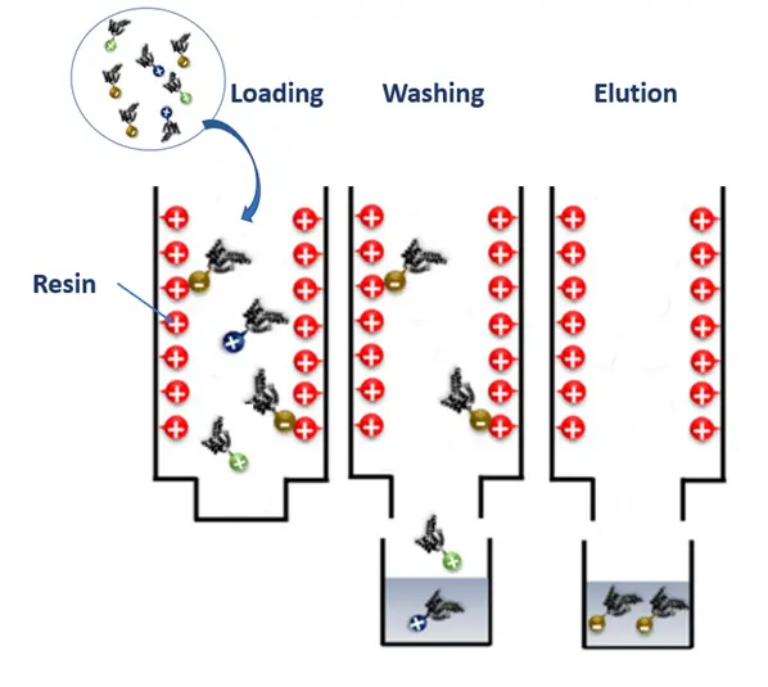
Ion exchange chromatography is an adsorption chromatography that relies on the electrostatic
interaction between the molecule or biomolecule to be purified and groups of opposite charge
covalently bound to the matrix.
Proteins are composed of amino acid sequences by which they acquire their charges. However, at a certain pH-Isoelectric Point, the net electrical charge of the protein is equal to zero. This parameter is of great importance as it allows the determination of the pH of the buffer used in the IEX protein separation. Therefore, to allow a protein separation based on their charge, the pH buffer should either be higher or inferior to the pI of the protein of interest.
Like all liquid chromatography techniques, IEX is composed of a mobile phase and a stationary phase:
Immobilized particles are always oppositely charged to the protein of interest. To this extent, two IEX types can be distinguished; Cation Exchange Chromatography (CEX), and Anion Exchange
Chromatography (AEX).
| IEX | Cation Exchange Chromatography | Anion Exchange Chromatography |
|---|---|---|
| Protein Adsorption | Positively charged protein | Negatively charged protein |
| pH Buffer | pH Buffer < pI | pH Buffer > pI |
Struggling with protein purification? Discover our full protein expression services!
