Cart (0 Items)
Your cart is currently empty.
View ProductsTagged protein purification is a robust protein purification method. It relies on the interaction between a fusion protein (Tag + Target protein) and its specific ligand immobilized on the resin. This method is usually employed to obtain highly pure protein solutions and to concentrate proteins present at low concentrations in complex mixtures.
Please note that tagged protein purification cannot be ordered as a standalone service but can be part of our protein expression services.

Benefit from our full protein expression services now! Contact us to help you choose the best protein purification method for your project.
Tag-affinity purification is one of the most common protein purification techniques. The former is based on the high affinity created between the fusion protein and its specific ligand. This method grants important purity levels. For instance, over 90% is easily achievable with this technique. Much higher purity percentages can even be achieved when other purification techniques are combined.
Affinity purification tags simplify the downstream processing into a single-step purification and increases yields through the specific interactions between ligands and fusion proteins. There are several types of protein tags. Yet, not all tags are suitable for all protein types and all downstream applications.
These tag types are small in size and rarely alter the protein’s structure and function. They allow specific protein separation from a complex protein mixture. To mention some examples:
In addition to their purification properties, solubility tags protect tagged proteins from proteolysis and enhance their solubility. However, their larger size adds more complexity to the downstream process. It is therefore important to cleave the tag at the end of the purification and eliminate the risk of altering the natural conformation of the protein of interest.
They are short amino acid sequences that rarely interfere with the protein conformation and activity. Epitopes serve for antibody purification and protein detection.
Affinity chromatography is a biochemical purification technique. It relies on reversible interactions between a protein and his ligand. The goal behind this technique is to isolate proteins of interest specifically from all other molecules allowing their purification from a heterogenous solution.
This type of chromatography is generally composed of a mobile phase and a stationary phase.
3 major steps compose affinity chromatography process:
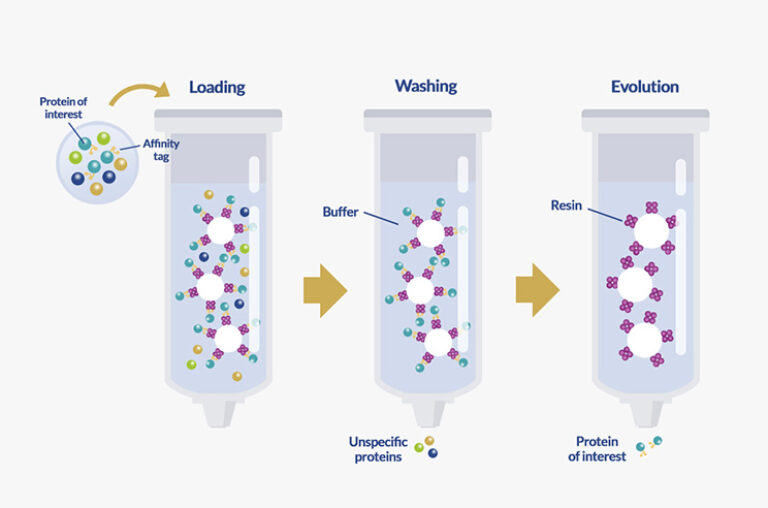
Successful protein purification by affinity chromatography relies highly on the tags and ligands used. The latter are responsible for the affinity to the proteins. Moreover, they provide specific interactions and stability to the process.
Here are most of the common affinity purification techniques:
Protein tags can vary from a short peptide sequence to a protein. They are synthesized along with the protein of interest in a way to be fused during the expression. Protein tags are mainly used to promote the protein purification process. Tags constitute an attractive approach as they simplify the downstream process into a single-step purification process. They can also serve in several other applications like protein characterization, and interaction studies. And bring in further advantages to the protein such as solubility and stability.
| Tag Type | Tag Name | Origin | Size (aa) | Interfering | Localization | Matrix |
|---|---|---|---|---|---|---|
| Affinity | Poly-Histidine | Synthetic | ~6 | Rarely | N-terminal C- terminal |
Immobilized metal ions: Ni2+, Co2+, Cu2+, Zn2+. |
| Affinity | Strep II-tag | Bacteria | 8 | No | N-terminal C- terminal |
Streptactin |
| Affinity | Poly-Arginine | Synthetic | 5 | Yes | C- terminal | Cation exchange resin. |
| Affinity | Calmodulin Binding Protein- CBP | Skeletal muscle myosin light chain kinase | 26 | No | N-terminal C- terminal |
Immobilized calmodulin. |
| Solubility | Glutathione-S-Transferase-GST | S.japonicum | 211 | Yes | N-terminal | Glutathione resin. |
| Solubility | Maltose-Binding Protein-MBP | E.coli | 396 | Yes | N-terminal C- terminal |
Immobilized amylose or maltose resins. |
| Solubility | Small Ubiquitin-Like-Modifier- SUMO | H.sapiens | ~100 | No | N-terminal | Catalytic core of Ulp1. |
| Solubility | N-utilization substance-NusA | E.coli | 495 | Yes | N-terminal C- terminal |
N/A |
| Epitope | FLAg tag Peptide- FLAG | Synthetic | 8 | Yes | N-terminal C- terminal |
Anti-FLAG antibody. |
| Epitope | c-MYC | H.sapiens | 10 | N/A | N-terminal C- terminal |
9E10 antibody coupled with agarose. |
| Epitope | EPEA / C-tag | Synthetic | 4 | Minimal | C-terminal | Erbin PDZ domain and immobilized on the agarose beads. |
| Epitope | Hemagglutinin (HA) | Human influenza HA molecule | 31 | N/A | N-terminal C- terminal |
Monoclonal antibodies. |
| Fluorescence | Green Fluorescent Protein (GFP) | Aequorea victoria | 220 | Yes | N-terminal C- terminal |
N/A |
| Fluorescence | S-tag | RNase A | 15 | No | N-terminal C- terminal |
S-fragment of RNase A. |
| Fluorescence | mCherry | DsRed of Discosoma | 256 | N/A | C- terminal | Affinity purified by Protein A. |
Thanks to immobilized antibodies, IAC can achieve highly specific purification. This technique is usually employed to identify and purify antigens and viral proteins. On the other hand, IAC can also serve for antibody purification, by replacing immobilized antibodies with immobilized antigens or proteins. In this context, protein A/G constitutes a popular choice for antibody purification.
To summarize, tagged protein purification offers affinity, specificity, and a fast purification process.
