Cart (0 Items)
Your cart is currently empty.
View Products
Expedite your therapeutic antibody development projects with our binder-guaranteed approach to antibody phage display services. Our high-diversity human (scFv and Fab) and camelid (VHH) libraries, allied to our expertise in collecting antibody repertoires in over 60 projects, ensures you always receive at least 3 unique functional binders. Drawing from our experience in 300+ phage display projects, we strive to help you create highly functional antibodies even when pursuing complex clinical targets.
An ideal example of the insights you can expect from this type of report
This comprehensive report includes:
Our fully human antibody library was designed to forgo time-consuming humanization processes
You get the full ownership of the antibody sequence generated!
Always receive AT LEAST 3 unique binders against your antigen! This is made possible thanks to the exceptional diversity of our libraries (>1010)
The variety of our libraries allows us to propose a wide range of formats suitable for therapeutic applications
From antigen to antibody in as little as 7 weeks
Minimize animal use in your antibody discovery projects by choosing our premium naïve libraries built for the highest possible quality and diversity
Oligoclonal therapies are ideal to tackle complex disease scenarios such as infectious diseases and envenoming
| Library | Format | Species | Size (clones) |
|---|---|---|---|
| NEW!
LiAb-SFCOVID-19TM |
scFv | Human – Donors that recovered from COVID-19 – Ideal for COVID-19 antibody generation | 1.19 X 1010 |
| LiAb-SFMAXTM | scFv & Fab | Human – 5 different ethnic groups – 368 donors for maximized diversity | 5.37 X 1010 |
| LiAb-SFaTM | scFv | Human | 1.5 X 109 |
| LiAb-FabTM | Fab | Human | 2.00 X 1010 |
| NEW! LiAb-VHHMAXTM | VHH | Camel, llama, alpaca – 57 animals – Ideal for NANOBODY® protein* generation | 1.51 X 1010 |
ProteoGenix’s proprietary libraries of fully human antibodies take your therapeutic development one step further by saving you the need to pursue antibody humanization and offering one of the most time-effective solutions to therapeutic antibody development. The high clonal and format diversity allied to the screening power of our phage display platform, are brought together to quick-start preclinical and clinical antibody development.
Fully human antibodies have been taking the lead in therapeutic applications due to their low immunogenicity and ability to engage patients’ immune systems for stronger responses. But NANOBODY® structures* have also been quickly gaining ground as biotherapeutics due to their astounding diffusion rates and ability to target cryptic epitopes that have remained elusive to conventional monoclonal antibodies.
Immune library
Antigen procurement or design and production
Immune library construction
Naive library
Antigen procurement or design and production
Library screening and biopanning
ELISA screening of single phage binders
DNA extraction & antibody sequencing
Affinity, selectivity (or cross-reactivity), and developability are three of the most important characteristics of successful therapeutic antibodies. Unlike other methods, phage display allows precise control over which specific epitopes are recognized by a certain antibody (selectivity), a faster way to screen for antibody affinity, and easier access to antibody sequences vital to evaluate antibody developability both in silico and in vitro. This early evaluation remains one of the most critical steps to ensure a smooth transition to large-scale production.
Other advantages of phage display platforms are tied to the nature of the antigen. It is well-documented that most clinical targets are membrane-bound proteins with complex conformations, unstable under standard conditions, and hard to express in recombinant systems. Since biopanning can be performed using immobilized antigens or in liquid suspensions, phage display can be adapted to these complex clinical targets ensuring the identification of the most biologically relevant binders.
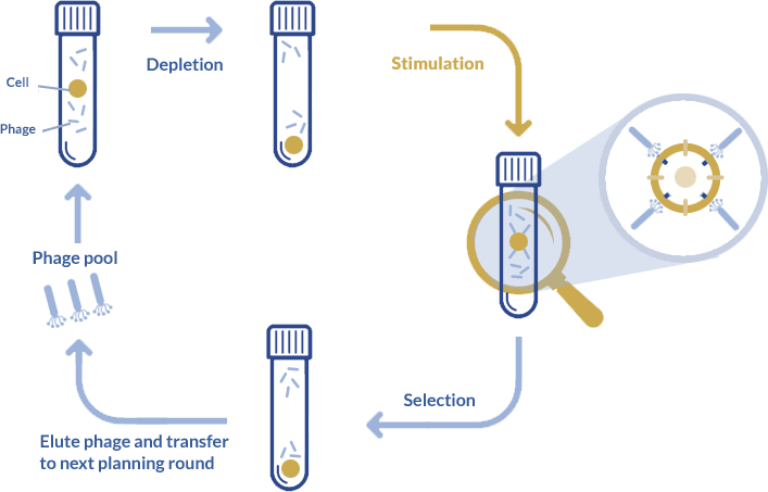
Adapted from Alfaleh, M. A. et al. Strategies for Selecting Membrane Protein-Specific Antibodies using Phage Display with Cell-Based Panning. Antibodies. 2017; 6(3):10. doi: 10.3390/antib6030010
The use of suspension cell-based antibody phage display continues to gain ground over other approaches to antibody phage display for therapeutic applications. This technique distinguishes itself from its ability to reproduce the cell environment as accurately as possible. Cells overexpressing the antigen on their surface thus ensure that only naturally exposed epitopes are recognized by these antibodies which increases the chances of identifying the most relevant binders.
Additionally, the use of phage display coupled with naïve antibody libraries ensures only a reduced amount of antigen is necessary to perform multiple biopanning campaigns, which ultimately reduces production costs and minimizes protein production hurdles.
Every project and clinical target are unique and require a detailed case-by-case analysis. At ProteoGenix, we believe this preliminary study dictates the success of each therapeutic antibody development project. The initial analysis allows us to predict and prepare for potential development issues as early as possible and decide on the best antibody generation approach: naïve versus immune.
For therapeutic antibody development, the use of antibody libraries generated from naïve hosts presents important advantages. These libraries comprise the highest possible antibody diversity which can be particularly beneficial for large projects developing antibodies against different antigens. Additionally, naïve libraries ensure early development timelines are kept to a minimum without sacrificing antibody quality and developability, vital when designing fast response biotherapeutics.
For more complex therapeutic targets, immune library development might be the best solution. With no species (except human) nor antigen restriction, ProteoGenix can boost the affinity of antibody repertoires by immunizing specific hosts and isolating relevant binders via library generation and phage display technologies. Despite the potentially higher affinity of immune library-derived antibodies, this approach is not without its challenges. Antibody library development can take up to 11 to 16 weeks due to the additional immunization and library construction step, significantly increasing early development timelines.
Anticancer monoclonal antibodies have been the leading biotherapeutics on the market. Cancer is a complex disease requiring a complex response. In this context, monoclonal antibodies quickly gained clinical interest due to their dual properties as antigen-binding and immune system engaging molecules.
However, cancer immunotherapies were not the only ones benefiting from the screening power of phage display technologies. Recent studies indicate phage display offers unique advantages for the development of next-generation envenoming treatments. Conventional anti-venom therapies have long depended on polyclonal sera generated in large mammals like horses. But pAbs have always suffered from well-documented drawbacks including batch-to-batch variability and immunogenicity greatly limiting their therapeutic efficacy.
Due to the versatility of phage display, it can be tailored to tackle the problem imposed by anti-venom development: the need to neutralize multiple different toxins at the same time. When high-diversity phage display libraries are used, biopanning can be performed easily against a panel of different toxins without the need for animal immunization (naïve antibody libraries). This ensures either high-quality antibody cocktails (a mixture of different monoclonal antibodies targeting different epitopes or antigens) or cross-reactive antibodies can be successfully developed for anti-toxin treatments.
Over the years, phage display has gained recognition as an efficient tool for therapeutic antibody discovery. But its usefulness extends beyond the early discovery stages. The technology continues to grow in popularity as the tool of choice for antibody lead optimization and engineering.
Allied to site-directed or random mutagenesis, phage display can be used to screen large pools of mutant variants to quickly identify binders with increased affinity. This approach has been recurrently applied to boost the affinity of weak antibodies and, more recently, to improve the production of bispecific antibodies. The underlying principle between the two approaches is the same: improve antibody binding properties.
In typical affinity maturation projects, specific residues from the complementarity-determining regions (CDRs) are mutated to enhance the affinity between this region and its specific antigen. In bispecific antibody engineering projects, regions outside the CDRs are typically selected to increase the affinity between antibody light and heavy chains in to ensure a higher affinity between the two and subsequent better assembly of the bispecific format without compromising antigen-binding affinity.
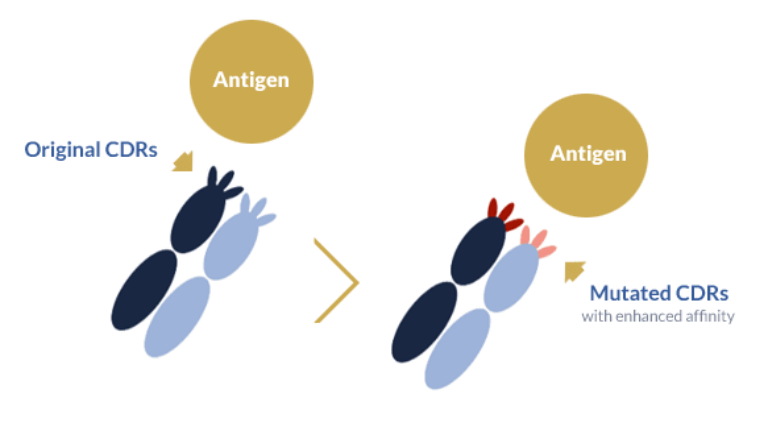
Phage display-based affinity maturation

Phage display-based bispecific antibody engineering
*NANOBODY® compound is a registered trademark of Ablynx N.V.
