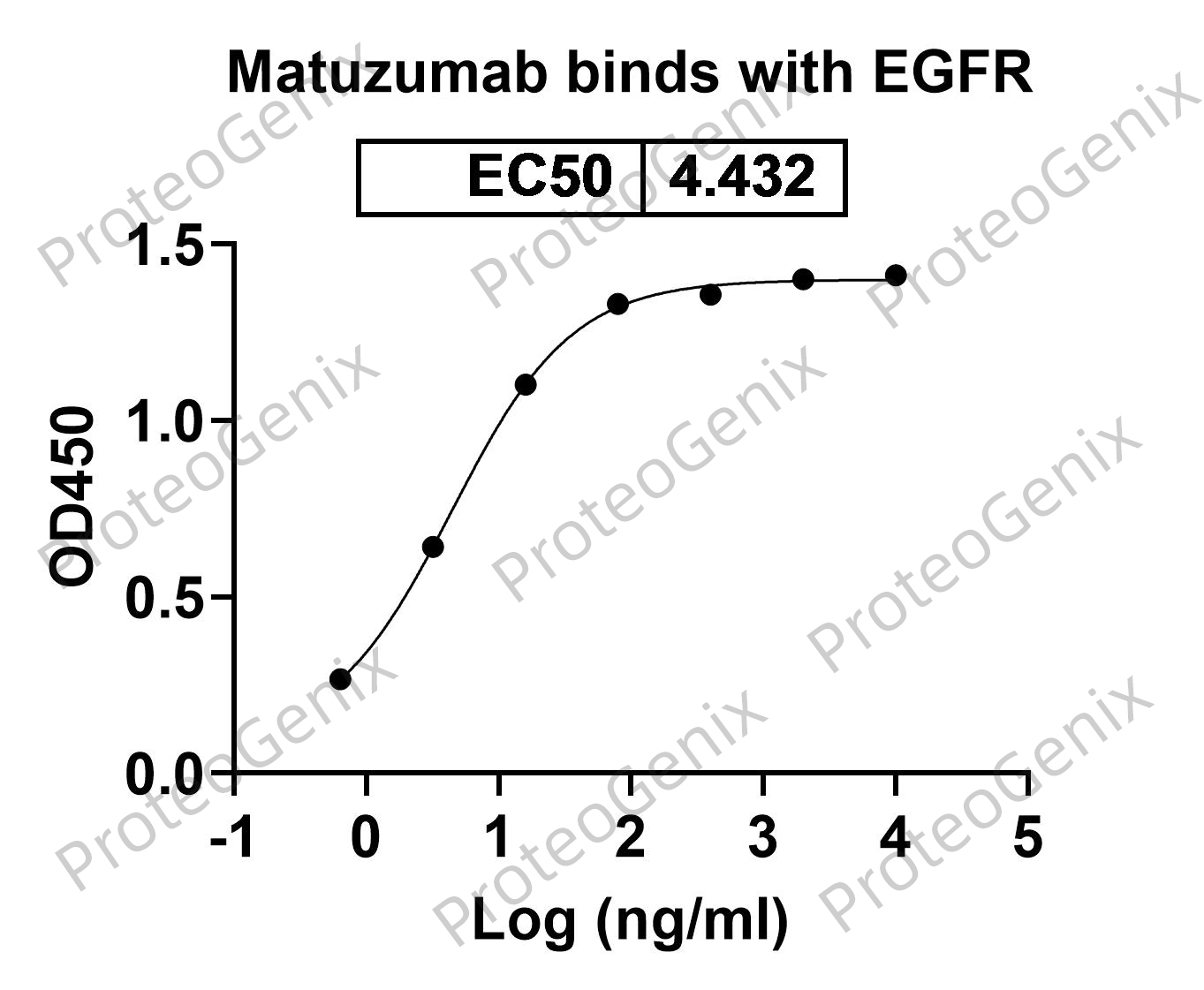
Introduction
Matuzumab Biosimilar, also known as Anti-EGFR mAb, is a research grade monoclonal antibody that has been developed as a potential therapeutic agent for various types of cancer. This biosimilar is designed to target the epidermal growth factor receptor (EGFR), a protein that is overexpressed in many types of cancer and plays a key role in tumor growth and progression.
Structure of Matuzumab Biosimilar
Matuzumab Biosimilar is a recombinant humanized monoclonal antibody that is produced through genetic engineering techniques. It is composed of two identical heavy chains and two identical light chains, each containing a variable region and a constant region. The variable region is responsible for binding to the target, while the constant region is responsible for mediating effector functions.
The variable region of Matuzumab Biosimilar is derived from a mouse monoclonal antibody that specifically binds to the extracellular domain of EGFR. This region has been humanized, meaning that it has been modified to have a structure similar to a human antibody, in order to reduce the risk of immune reactions when administered to patients.
Mechanism of Action
Matuzumab Biosimilar works by binding to the extracellular domain of EGFR, preventing the binding of epidermal growth factor (EGF) and other ligands. This blocks the activation of downstream signaling pathways that promote cell proliferation and survival, ultimately leading to the inhibition of tumor growth.
In addition, Matuzumab Biosimilar can also induce antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). This means that it can recruit immune cells and activate the complement system to directly kill cancer cells that express EGFR.
Applications of Matuzumab Biosimilar
Matuzumab Biosimilar is being investigated as a potential treatment for various types of cancer, including colorectal, lung, and head and neck cancers. It is also being studied in combination with other anti- cancer therapies, such as chemotherapy and radiation, to enhance its efficacy.
One of the key advantages of Matuzumab Biosimilar is that it specifically targets EGFR, which is overexpressed in many types of cancer. This makes it a promising therapeutic option for patients who have tumors with high levels of EGFR expression, as it can potentially have a more targeted and effective treatment compared to traditional chemotherapy.
Research Grade vs. Clinical Grade
Matuzumab Biosimilar is currently only available in a research grade, meaning that it is intended for use in laboratory and preclinical studies. This allows researchers to evaluate its safety and efficacy in different cancer models before it can be further developed for clinical use.
In order for Matuzumab Biosimilar to be used in clinical settings, it will need to undergo further testing and obtain regulatory approval. This involves conducting clinical trials to demonstrate its safety and efficacy in human patients, as well as obtaining manufacturing and quality control processes to ensure consistency and purity of the product.
Conclusion
Matuzumab Biosimilar is a promising research grade monoclonal antibody that specifically targets EGFR and has the potential to be an effective treatment for various types of cancer. Its unique mechanism of action and potential for combination therapy make it a valuable asset in the fight against cancer. Further research and development is needed to bring this biosimilar to the clinical setting and provide a new treatment option for cancer patients.
Keywords: antibody, therapeutic target, Matuzumab Biosimilar, Anti-EGFR mAb, research grade, structure, activity, application, cancer, EGFR, mechanism of action, ADCC, CDC, clinical grade.
There are no reviews yet.